Advertisements
Advertisements
प्रश्न
Describe the roles of the following: SiO2 in the extraction of copper from copper matte
Advertisements
उत्तर
In the extraction of copper from copper matte, SiO2 is used to remove iron oxide as slag
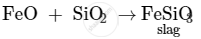
APPEARS IN
संबंधित प्रश्न
Draw neat and labelled diagram of Bessermer converter used in the extraction of copper.
What is the role of CO in the extraction of Iron?
What is the role of coke in the extraction of iron from its oxides?
Name the method used for refining of copper metal.
Which among the following minerals does NOT contain aluminium?
(a) Cryolite
(b) Siderite
(c) China clay
(d) Corundum
Draw a neat, well labelled diagram of electrolytic cell for extraction of aluminium
Carbonate ore of iron is ____________.
In the electrolytic process for the extraction of aluminium the electrolyte is:-
In the extraction of iron, the slag produce is
Calamine and Malachite, respectively, are the ores of ______.
