Advertisements
Advertisements
Question
Describe the roles of the following: SiO2 in the extraction of copper from copper matte
Advertisements
Solution
In the extraction of copper from copper matte, SiO2 is used to remove iron oxide as slag
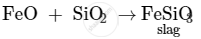
APPEARS IN
RELATED QUESTIONS
Write the names and chemical formulae of any one ore of zinc.
Write chemical formulae of the following ores :
(a) Calamine
(b) Haematite
(c) Magnetite
(d) Corundum
Fe2O3 is reduced to spongy iron near the top of blast furance by
(A) H2
(B) CaO
(C) SiO2
(D) CO
Draw neat and labelled diagram of Bessermer converter used in the extraction of copper.
What is the role of CO in the extraction of Iron?
Indicate the principle behind the method used for the refining of zinc.
Copper has the face centred cubic structure. The coordination number of each ion is:
Give the uses of zinc.
When a strip of metallic zinc is placed in an aqueous solution of copper nitrate the blue colour of the solution disappear due to formation of
In the thermite process, the reducing agent is:
