Advertisements
Advertisements
प्रश्न
An organic compound (A) with molecular formula C3H7NO on heating with Br2 and KOH forms a compound (B). Compound (B) on heating with CHCl3 and alcoholic KOH produces a foul-smelling compound (C) and on reacting with C6H5SO2Cl forms a compound (D) which is soluble in alkali. Write the structure of (A), (B), (C) and (D).
Advertisements
उत्तर
\[\ce{CH3\underset{(A)}{CH2}CONH2 + Br2 + KOH -> CH3\underset{(B)}{CH2}NH2}\]
\[\ce{CH3\underset{(B)}{CH2}NH2 + CHCl3 + KOH -> CH3\underset{(C)}{CH2}NC + KCl + H2O}\]

A = Propanaimde
B = Ethyl amine (1° amine)
C = Ethyl isocyanide
D = N-ethyl benzene sulphonamide
APPEARS IN
संबंधित प्रश्न
Write the IUPAC name of the following compound and classify it as primary, secondary and tertiary amine.
CH3(CH2)2NH2
Give one chemical test to distinguish between the following pair of compounds.
Methylamine and dimethylamine
Give one chemical test to distinguish between the following pair of compounds.
Aniline and N-methylaniline
How will you convert ethanoic acid into methanamine?
How will you convert hexanenitrile into 1-aminopentane?
How will you convert nitromethane into dimethylamine?
Do the following conversions in not more than two steps :
Propanone to Propene
Write the IUPAC name of the given compound :
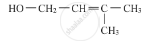
Identify the incorrect IUPAC name.
Do the following conversions in not more than two steps:
\[\begin{array}{cc}
\ce{CH3CN to CH3 - C - CH3}\\
\phantom{...........}||\\
\phantom{...........}\ce{O}
\end{array}\]
