Advertisements
Advertisements
प्रश्न
Do the following conversions in not more than two steps:
\[\begin{array}{cc}
\ce{CH3CN to CH3 - C - CH3}\\
\phantom{...........}||\\
\phantom{...........}\ce{O}
\end{array}\]
Advertisements
उत्तर
\[\begin{array}{cc}
\phantom{.............................}\ce{Cl}\\
\phantom{.................................}\backslash\\
\ce{CH3 - C ≡ N + CH3MgCl -> CH3 - C = NMgCl + 2H2O ->[H+] Mg + NH3 + CH3 -C - CH3}\\
\phantom{................................}|\phantom{.......................}/\phantom{....................}||\phantom{..}\\
\phantom{..............................}\ce{CH3}\phantom{.................}\ce{OH}\phantom{.....................}\ce{O}\\\\\\\phantom{.}\\
\end{array}\]
संबंधित प्रश्न
Write the IUPAC name of the following compound and classify it as primary, secondary and tertiary amine.
(CH3)2CHNH2
Write the IUPAC name of the following compound and classify it as primary, secondary and tertiary amine.
(CH3CH2)2NCH3
Give one chemical test to distinguish between the following pair of compounds.
Ethylamine and aniline
How will you convert ethanoic acid into propanoic acid?
Accomplish the following conversion:
Chlorobenzene to p-chloroaniline
Complete the following reaction:
\[\ce{C6H5NH2 + H2SO4 (conc.)}\]
What is the action of p-toluenesulphonychloride on ethylamine and diethylamine?
Write the IUPAC name of the given compound :
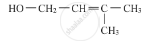
Identify the incorrect IUPAC name.
Do the following conversions in not more than two steps:

