Advertisements
Advertisements
प्रश्न
Give one chemical test to distinguish between the following pair of compounds.
Ethylamine and aniline
Advertisements
उत्तर
Ethylamine is a primary aliphatic amine, while aniline is a primary aromatic amine. They can be differentiated by azo dye test.
Azo dye test: In this, aromatic primary amine reacts with HNO2 (NaNO2 + dil. HCl) at 273 – 278 K and then reacts with alkaline solution of 2-naphthol (β-naphthol) to give a deep yellow, orange or red colored dye.
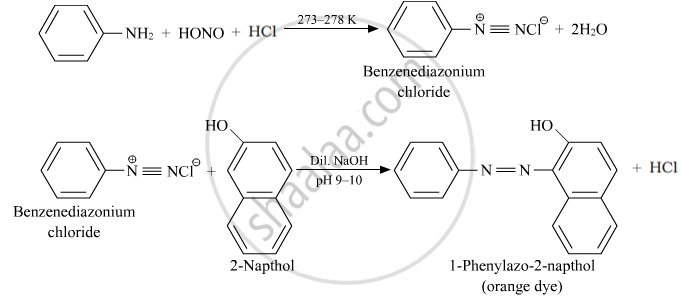
Aliphatic primary amines under the above conditions rapidly release nitrogen gas with the formation of primary alcohols, i.e., the solution remains transparent.
\[\ce{\underset{Ethylamine}{C2H5NH2} + HONO ->[273 - 278 K] \underset{ethyl alcohol}{C2H5OH} + H2O + N2 ^}\]
APPEARS IN
संबंधित प्रश्न
Write the IUPAC name of the following compound and classify it as primary, secondary and tertiary amine.
CH3NHCH(CH3)2
Write the IUPAC name of the following compound and classify it as primary, secondary and tertiary amine.
(CH3)3CNH2
Write the IUPAC name of the following compound and classify it as primary, secondary and tertiary amine.
(CH3CH2)2NCH3
Give one chemical test to distinguish between the following pair of compounds.
Methylamine and dimethylamine
Give one chemical test to distinguish between the following pair of compounds.
Secondary and tertiary amines
How will you convert ethanoic acid into methanamine?
How will you convert propanoic acid into ethanoic acid?
Accomplish the following conversion:
Aniline to 2, 4, 6-tribromofluorobenzene
Accomplish the following conversion:
Aniline to benzyl alcohol
An aromatic compound ‘A’ on treatment with aqueous ammonia and heating forms compound ‘B’ which on heating with Br2 and KOH forms a compound ‘C’ of molecular formula C6H7N. Write the structures and IUPAC names of compounds A, B and C.
Complete the following reaction:
\[\ce{C6H5N2Cl + C2H5OH ->}\]
Complete the following reaction:
\[\ce{C6H5NH2 + Br2 (aq) ->}\]
Complete the following reaction:
\[\ce{C6H5NH2 + (CH3CO)2O ->}\]
How are ethylamine and ethyl methyl amine distinguished by using nitrous acid?
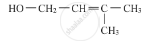
Write the IUPAC name of the given compound :
Using IUPAC norms, write the formula of Hexaamminecobalt(III) sulphate.
