Advertisements
Advertisements
प्रश्न
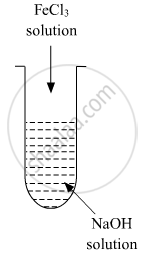
A colloidal sol is prepared by the given method in the figure. What is the charge on hydrated ferric oxide colloidal particles formed in the test tube? How is the sol represented?

Advertisements
उत्तर
Colloidal particles usually adsorb those ions which are in excess and are common to its own lattice. This preferential adsorption of a particular type of ions imparts a particular type of charge to colloidal particles.
When FeCl3 is added in NaOH, the constituent of the sol is Fe(OH)3 but the dispersion medium is having an excess of OH- ions. Hence, it gets preferentially adsorbed to the sol giving the overall negative charge. The sol is represented as Fe2O3.xH2O / OH-
APPEARS IN
संबंधित प्रश्न
Define the following terms: Associated colloids
Explain what is observed when an electric current is passed through a colloidal sol?
Explain the terms Coagulation
Which phenomenon occurs when an electric field is applied to a colloidal solution and electrophoresis is prevented?
(i) Reverse osmosis takes place.
(ii) Electroosmosis takes place.
(iii) Dispersion medium begins to move.
(iv) Dispersion medium becomes stationary.
How does it become possible to cause artificial rain by spraying silver iodide on the clouds?
The important factor in the stabilization of a colloidal solution is ______
Which of the following is a correct statement?
The conditions given below are in the context of observing Tyndall effect in colloidal solutions:
- The diameter of the colloidal particles is comparable to the wavelength of light used.
- The diameter of the colloidal particles is much smaller than the wavelength of light used.
- The diameter of the colloidal particles is much larger than the wavelength of light used.
- The refractive indices of the dispersed phase and the dispersion medium are comparable.
- The dispersed phase has a very different refractive index from the dispersion medium.
Choose the most appropriate conditions from the options given below:
Coagulation value of the electrolytes AlCl3 and NaCl for As2S3 sol are 0.093 and 52 respectively. The No. of times AlCl3 has greater coagulating power than NaCl is ______.
6.84 g Al2(SO4)3 is needed to coagulate 2.5 L of As2S3 sol completely in 2.0 hrs. The coagulation value of Al2 (SO4)3 is ______.
