Advertisements
Advertisements
प्रश्न
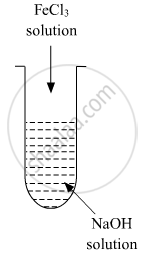
A colloidal sol is prepared by the given method in the figure. What is the charge on hydrated ferric oxide colloidal particles formed in the test tube? How is the sol represented?

Advertisements
उत्तर
Colloidal particles usually adsorb those ions which are in excess and are common to its own lattice. This preferential adsorption of a particular type of ions imparts a particular type of charge to colloidal particles.
When FeCl3 is added in NaOH, the constituent of the sol is Fe(OH)3 but the dispersion medium is having an excess of OH- ions. Hence, it gets preferentially adsorbed to the sol giving the overall negative charge. The sol is represented as Fe2O3.xH2O / OH-
APPEARS IN
संबंधित प्रश्न
Define the following terms: Associated colloids
Out of BaCl2 and KCl, which one is more effective in causing coagulation of a negatively charged colloidal Sol? Give reason.
What happens when an emulsion is centrifuged?
A colloidal sol is prepared by the given method in the figure. What is the charge on hydrated ferric oxide colloidal particles formed in the test tube? How is the solution represented?

The values of colligative properties of colloidal solution are of small order in comparison to those shown by true solutions of same concentration because of colloidal particles ______.
Why do we add alum to purify water?
Assertion: Colloidal solutions do not show brownian motion.
Reason: Brownian motion is responsible for stability of sols.
Gold number of protective colloids A, Band Dare 0.50, 0.01 and 0.10 and 0.005. The correct order of their protecting power is
The right option for the statement “Tyndall effect is exhibited by”, is:
Coagulation value of the electrolytes AlCl3 and NaCl for As2S3 sol are 0.093 and 52 respectively. The No. of times AlCl3 has greater coagulating power than NaCl is ______.
