Please select a subject first
Advertisements
Advertisements
Draw shapes of 2p orbitals.
Concept: undefined >> undefined
Explain in brief, the significance of the azimuthal quantum number.
Concept: undefined >> undefined
Advertisements
If n = 3, what are the quantum number l and m?
Concept: undefined >> undefined
The electronic configuration of oxygen is written as 1s2 2s2 \[\ce{2p^2_{{x}}}\] \[\ce{2p^1_{{y}}}\] \[\ce{2p^1_{{z}}}\] and not as 1s2 2s2 \[\ce{2p^2_{{x}}}\], \[\ce{2p^2_{{y}}}\] \[\ce{2p^0_{{z}}}\], Explain.
Concept: undefined >> undefined
Write a note on ‘Principal Quantum number.
Concept: undefined >> undefined
Using the concept of quantum numbers, calculate the maximum numbers of electrons present in the ‘M’ shell. Give their distribution in shells, subshells, and orbitals.
Concept: undefined >> undefined
Indicate the number of unpaired electrons in \[\ce{Si}\] (Z = 14).
Concept: undefined >> undefined
Indicate the number of unpaired electron in:
Cr (Z = 24)
Concept: undefined >> undefined
Choose the correct option.
Colourless components on chromatogram can not be observed by the following:
Concept: undefined >> undefined
Give names of two materials used as stationary phase in chromatography.
Concept: undefined >> undefined
Define the Stationary phase.
Concept: undefined >> undefined
Name the different types of Chromatography and explain the principles underlying them.
Concept: undefined >> undefined
How do you visualize colourless compounds after separation in TLC and Paper Chromatography?
Concept: undefined >> undefined
Label the diagram and explain the process in your words.
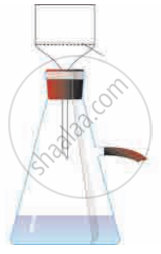
Concept: undefined >> undefined
Compare TLC and Paper Chromatography techniques.
Concept: undefined >> undefined
Define the term Inhibition.
Concept: undefined >> undefined
Define the term Catalysis.
Concept: undefined >> undefined
Mention various applications of equilibrium constant.
Concept: undefined >> undefined
Identify nuclear fusion reaction.
Concept: undefined >> undefined
Choose the correct option.
The process by which nuclei having low masses are united to form nuclei with large masses is ______.
Concept: undefined >> undefined
