Advertisements
Advertisements
प्रश्न
When a Coolidge tube is operated for some time it becomes hot. Where does the heat come from?
Advertisements
उत्तर
A Coolidge tube apparatus consists of a filament and a target. The filament is heated to produce electrons that are accelerated by applying an electric field between the filament and the target. When these accelerated electrons enter the target, they collide with the target atoms. In the process, the electrons lose their kinetic energy. A part of this kinetic energy is utilised for emitting X-rays and the remaining energy is absorbed by the target. Inside the target, the kinetic energy of the electrons is converted into heat energy. This raises the temperature of the target and hence, it heats the Coolidge tube.
APPEARS IN
संबंधित प्रश्न
What role dose infra-red radiation play in maintain the Earth’s warmth
Identify the part of the electromagnetic spectrum which is:
produced by bombarding a metal target by high speed electrons.
Name the electromagnetic radiations used for (a) water purification, and (b) eye surgery.
Name the subjective property of light related to its wavelength.
Two waves A and B have wavelength 0.01 Å and 9000 Å respectively.
- Name the two waves.
- Compare the speeds of these waves when they travel in vacuum.
X-ray and visible light travel at the same speed in vacuum. Do they travel at the same speed in glass?
Consider a photon of a continuous X-ray coming from a Coolidge tube. Its energy comes from
Moseley's Law for characteristic X-ray is √v = a(Z − b). Here,
50% of the X-ray coming from a Coolidge tube is able to pass through a 0.1 mm thick aluminium foil. The potential difference between the target and the filament is increased. The thickness of the aluminium foil that will allow 50% of the X-ray to pass through will be
When 40 kV is applied across an X-ray tube, X-ray is obtained with a maximum frequency of 9.7 × 1018 Hz. Calculate the value of Planck constant from these data.
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
A free atom of iron emits Kα X-rays of energy 6.4 keV. Calculate the recoil kinetic energy of the atom. Mass of an iron atom = 9.3 × 10−26 kg.
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
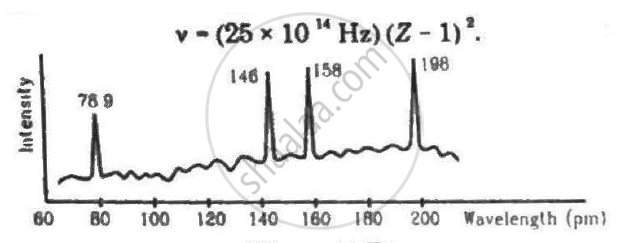
Continuous X-rays are made to strike a tissue paper soaked with polluted water. The incoming X-rays excite the atoms of the sample by knocking out the electrons from the inner shells. Characteristic X-rays are analysed and the intensity is plotted against the wavelength. Assuming that only Kα intensities are detected, list the elements present in the sample from the plot. Use Moseley's equation v − (25 × 1014Hz)(Z − 1)2.
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
| Gamma rays | D | C | Visible light | B | A |
The above table shows different parts of the electromagnetic spectrum.
(a) Identify the parts of the spectrum marked as A, B, C and D.
(b) Which of the radiations A or B has the higher frequency?
(c) State two properties which are common to all parts of the electromagnetic spectrum.
(d) Name one source of each of the radiation of electromagnetic spectrum.
(e) Name one detector for each of the radiation.
(f) Name one use of each of the radiation.
Gamma rays and radio waves travel with the same velocity in free space. Distinguish between them in terms of their origin and the main application.
Choose the correct option.
Earth’s atmosphere is richest in
Answer briefly.
Why light waves travel in a vacuum whereas sound waves cannot?
An electron beam is accelerated by a potential difference V to hit a metallic target to produce X-rays. It produces continuous as well as characteristic X-rays. If λmin is the smallest possible wavelength of X-ray in the spectrum, the variation of log λmin with log V is correctly represented in:
All components of the electromagnetic spectrum in a vacuum have the same ______
In uranium (Z = 92) the K absorption edge is 0.107 Å and the Kα line is 0.126 Å, and the wavelength of the L absorption edge is ______.
What happens when an electron collides with a positron?
