Advertisements
Advertisements
प्रश्न
Choose the correct option.
Earth’s atmosphere is richest in
विकल्प
UV
IR
X-ray
Microwaves
Advertisements
उत्तर
Earth’s atmosphere is richest in IR
APPEARS IN
संबंधित प्रश्न
What physical quantity is the same for X-rays of wavelength 10−10 m, red light of wavelength 6800 Å and radiowaves of wavelength 500 m?
Give a reason for the following:
Long-distance radio broadcasts use short-wave bands. Why?
Optical and radio telescopes are built on the ground but X-ray astronomy is possible only from satellites orbiting the earth. Why?
What is the range of the wavelength of the following electromagnetic waves?
(a) Micro waves .
Give the range of wavelength of the electromagnetic waves visible to us.
Name the waves used for taking photographs in dark.
The wavelength of X-rays is 0.01 Å. Calculate its frequency. State the assumption made, if any.
Can Lα X-ray of one material have shorter wavelength than Kα X-ray of another?
Consider a photon of a continuous X-ray coming from a Coolidge tube. Its energy comes from
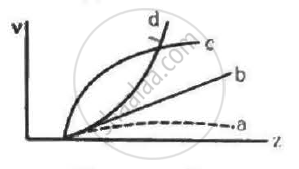
Frequencies of Kα X-rays of different materials are measured. Which one of the graphs in the figure may represent the relation between the frequency v and the atomic number Z ?
The potential difference applied to an X-ray tube is increased. As a result, in the emitted radiation,
(a) the intensity increases
(b) the minimum wavelength increases
(c) the intensity remains unchanged
(d) the minimum wavelength decreases.
Find the energy, the frequency and the momentum of an X-ray photon of wavelength 0.10 nm.
(Use Planck constant h = 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
The wavelength of Kα X-ray of tungsten is 21.3 pm. It takes 11.3 keV to knock out an electron from the L shell of a tungsten atom. What should be the minimum accelerating voltage across an X-ray tube having tungsten target which allows production of Kα X-ray?
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
The energy of a silver atom with a vacancy in K shell is 25.31 keV, in L shell is 3.56 keV and in M shell is 0.530 keV higher than the energy of the atom with no vacancy. Find the frequency of Kα, Kβ and Lα X-rays of silver.
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
When 40 kV is applied across an X-ray tube, X-ray is obtained with a maximum frequency of 9.7 × 1018 Hz. Calculate the value of Planck constant from these data.
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
Suppose a monochromatic X-ray beam of wavelength 100 pm is sent through a Young's double slit and the interference pattern is observed on a photographic plate placed 40 cm away from the slit. What should be the separation between the slits so that the successive maxima on the screen are separated by a distance of 0.1 mm?
Identify the part of the electromagnetic spectrum used in (i) radar and (ii) eye surgery. Write their frequency range.
Name any two electromagnetic waves which have a frequency higher than that of violet light. State one use of each.
Name two sources of infrared radiation.
Answer briefly.
Can we produce a pure electric or magnetic wave in space? Why?
What are the ultraviolet rays?
Answer briefly.
Name the most harmful radiation entering the Earth's atmosphere from outer space.
An e.m. wave exerts pressure on the surface on which it is incident. Justify.
A bat moving at 10 ms−1 towards a wall sends a sound signal of 8000 Hz towards it. On reflection, it hears a sound of frequency f The value of f in Hz is close to (speed of sound = 320 ms−1)
If λv, λx and λm Am represents the wavelength of visible light, x-ray and microwaves respectively, then ______.
The ratio of contributions made by the electric field and magnetic field components to the intensity of an EM wave is ______.
Photons of an electromagnetic radiation has an energy 11 keV each. To which region of electromagnetic spectrum does it belong?
Identify the electromagnetic wave whose wavelength range is from about 10-3 m to about 10-1 m. Write one use of this.
Choose the correct option related to wavelengths (λ) of different parts of the electromagnetic spectrum.
