Advertisements
Advertisements
प्रश्न
Using valence bond theory, explain the following in relation to the complexes given below:
\[\ce{[Mn(CN)6]^{3-}}\]
(i) Type of hybridisation.
(ii) Inner or outer orbital complex.
(iii) Magnetic behaviour.
(iv) Spin only magnetic moment value.
Advertisements
उत्तर
\[\ce{[Mn(CN)6]^{3-}}\]:
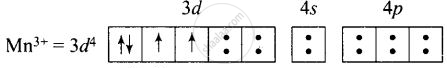
(i) Hybridisation – d2sp3
(ii) Inner orbital complex
(iii) Paramagnetic
(iv) Magnetic moment = `sqrt(2(2 + 2))` = 2.87 B.M
APPEARS IN
संबंधित प्रश्न
On the basis of valence bond theory explain the nature of bonding in [CoF6]3 ion.
[NiCl4]2− is paramagnetic, while [Ni(CO)4] is diamagnetic, though both are tetrahedral. Why? (Atomic number of Ni = 28)
Discuss the nature of bonding in the following coordination entity on the basis of valence bond theory:
[FeF6]3−
Discuss the nature of bonding in the following coordination entity on the basis of valence bond theory:
[Co(C2O4)3]3−
Explain the geometry of `[Co(NH_3)_6]^(3+)` on the basis of hybridisation. (Z of Co = 27)
Using valence bond theory, explain the following in relation to the complexes given below:
\[\ce{[FeCl6]^{4-}}\]
(i) Type of hybridisation.
(ii) Inner or outer orbital complex.
(iii) Magnetic behaviour.
(iv) Spin only magnetic moment value.
Write the hybridization and shape of the following complexes:
[Ni(CN)4]2−
In a coordination entity, the electronic configuration of the central metal ion is t2g3 eg1
Is the coordination compound a high spin or low spin complex?
If orbital quantum number (l) has values 0, 1, 2 and 3, deduce the corresponding value of principal quantum number, n.
How many radial nodes for 3p orbital?
When the hybridization state of carbon changes from sp3 to sp2 and finally to sp, the angle between hybridized orbital will
What is the no. of possible isomers for the octahedral complex [Co(NH3)2(C2O4)2]?
In Fe(CO)5, the Fe – C bond possesses
Valence bond theory is based on the assumption that the bonds formed between the metal ions and ligands are ______
Using Valence bond theory, explain the following in relation to the paramagnetic complex [Mn(CN)6]3−
- type of hybridization
- magnetic moment value
- type of complex – inner, outer orbital complex
Using valence bond theory, predict the hybridization and magnetic character of the following:
[CoF6]3– [Atomic number of Co = 27]
According to the valence bond theory, the hybridization of central metal atom is dsp2 for which one of the following compounds?
The magnetic moment of [NiCl4]2− is ______.
[Atomic number: Ni = 28]
[Ni(CO)4] has tetrahedral geometry while [Ni(CN)4]2− has square planar, yet both exhibit diamagnetism. Explain.
[Atomic number: Ni = 28]
