Advertisements
Advertisements
प्रश्न
Using crystal field theory, draw energy level diagram, write electronic configuration of the central metal atom/ion and determine the magnetic moment value in the following:
\[\ce{[FeF6]^{3-}, [Fe(H2O)6]^{2+}, [Fe(CN)6]^{4-}}\]
Advertisements
उत्तर
(1) \[\ce{FeF^{3-}6}\]:
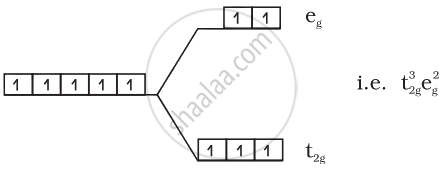
\[\ce{Fe^{3+} = 3d^5}\]
Number of unpaired electrons = 5
Magnetic moment = `sqrt(5(5 + 2))` = 5.92 BM
(2) \[\ce{[Fe(H2O)6]^{2+}}\]:
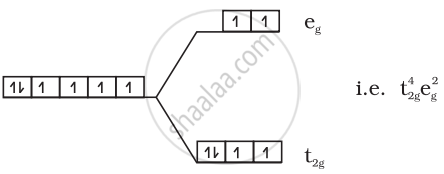
\[\ce{Fe^{2+} = 3d^6}\]
Number of unpaired electrons = 4
Magnetic moment = `sqrt(4(4 + 2))` = 4.9 BM
(3) \[\ce{[Fe(CN)6]^{4-}}\]:
\[\ce{Fe^{2+} = 3d^6}\]
Since CN– is a strong field ligand all the electrons get paired.
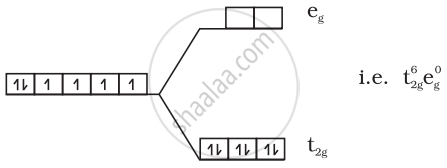
No unpaired electrons so diamagnetic.
APPEARS IN
संबंधित प्रश्न
On the basis of crystal field theory, write the electronic configuration for d4 ion if ∆0 < P.
The hexaquo manganese(II) ion contains five unpaired electrons, while the hexacyanoion contains only one unpaired electron. Explain using Crystal Field Theory.
How does the magnitude of Δ0 decide the actual configuration of d orbitals in a coordination entity?
Write the electronic configuration of Fe(III) on the basis of crystal field theory when it forms an octahedral complex in the presence of (i) strong field, and (ii) weak field ligand. (Atomic no.of Fe=26)
Complete and balance the following reactions:
\[\ce{P4 + H2SO4 ->}\] ______ + ______ + ______
The CFSE for octahedral \[\ce{[CoCl6]^{4-}}\] is 18,000 cm–1. The CFSE for tetrahedral \[\ce{[CoCl4]^{2-}}\] will be ______.
An aqueous pink solution of cobalt (II) chloride changes to deep blue on addition of excess of HCl. This is because:
(i) \[\ce{[Co(H2O)6]^{2+}}\] is transformed into \[\ce{[CoCl6]}^{4-}\]
(ii) \[\ce{[Co(H2O)6]^{2+}}\] is transformed into \[\ce{[CoCl4]}^{2-}\]
(iii) tetrahedral complexes have smaller crystal field splitting than octahedral complexes.
(iv) tetrahedral complexes have larger crystal field splitting than octahedral complex.
Why are low spin tetrahedral complexes not formed?
Arrange following complex ions in increasing order of crystal field splitting energy (∆O):
\[\ce{[Cr(Cl)6]^{3-}, [Cr(CN)6]^{3-}, [Cr(NH3)6]^{3+}}\].
Match the complex ions given in Column I with the hybridisation and number of unpaired electrons given in Column II and assign the correct code:
| Column I (Complex ion) | Column II (Hybridisation, number of unpaired electrons) |
| A. \[\ce{[Cr(H2O)6]^{3+}}\] | 1. dsp2, 1 |
| B. \[\ce{[Co(CN)4]^{2-}}\] | 2. sp3d2, 5 |
| C. \[\ce{[Ni(NH3)6]^{2+}}\] | 3. d2sp3, 3 |
| D. \[\ce{[MnF6]^{4-}}\] | 4. sp3, 4 |
| 5. sp3d2, 2 |
The CFSE for octahedral [CoCl6]−4 is 18,000 cm−1. What will be the CFSE for tetrahedral [CoCl3]−2?
Considering crystal field theory, strong-field ligands such as CN–:
The correct order of increasing crystal field strength in following series:
Crystal field stabilising energy for high spind4 octahedral complex is:-
What is the difference between a weak field ligand and a strong field ligand?
What is crystal field splitting energy?
The correct order of intensity of colors of the compounds is ______.
For octahedral Mn(II) and tetrahedral Ni(II) complexes, consider the following statements:
- Both the complexes can be high spin.
- Ni(II) complex can very rarely below spin.
- With strong field Ligands, Mn(II) complexes can be low spin.
- Aqueous solution of Mn (II) ions is yellow in colour.
The correct statements are:
The value of the spin only magnetic moment for one of the following configurations is 2.84 BM. The correct one is:
