Advertisements
Advertisements
प्रश्न
The free surface of oil in a tanker, at rest, is horizontal. If the tanker starts accelerating the free surface will be titled by an angle θ. If the acceleration is a ms–2, what will be the slope of the free surface?
Advertisements
उत्तर
The behaviour of a liquid contained in a horizontally accelerated vessel can be understood by understanding the behaviour of a pendulum suspended from the ceiling of a horizontally accelerated trolley.
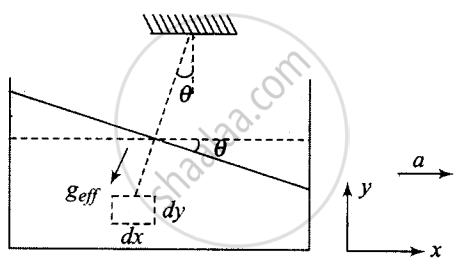
Every fluid element attains an equilibrium position under the action of gravity and pseudo-force. The free surface of the liquid orients itself perpendicular to the direction of net effective gravity.
tan θ = a/g
Suppose a tanker accelerates along the x-axis with acceleration a, the free surface of the tanker will not be horizontal because pseudo force acts as shown in the diagram.
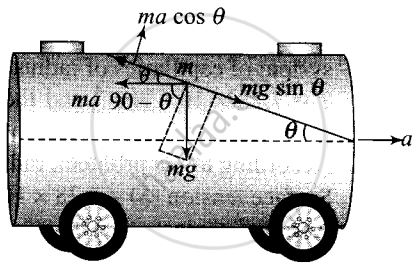
Consider an elementary particle of the oil of mass m.
The acting forces on the particle with respect to the tanker are shown in the figure alongside.
Now, balancing forces (as the particle is in equilibrium) along the inclined direction of the surface.
ma = pseudo force
mg = weight of small part of the oil.
Along the free surface,
Net force = 0
⇒ ma cos θ = mg sin θ
⇒ a = g tan θ
⇒ θ = tan-1(a/g)
APPEARS IN
संबंधित प्रश्न
Derive Laplace’s law for spherical membrane of bubble due to surface tension.
The contact angle between water and glass is 0°. When water is poured in a glass to the maximum of its capacity, the water surface is convex upward. The angle of contact in such a situation is more than 90°. Explain.
When water droplets merge to form a bigger drop
If two soap bubbles of different radii are connected by a tube,
The excess pressure inside a soap bubble is twice the excess pressure inside a second soap bubble. The volume of the first bubble is n times the volume of the second where n is
A capillary tube of radius 1 mm is kept vertical with the lower end in water. (a) Find the height of water raised in the capillary. (b) If the length of the capillary tube is half the answer of part , find the angle θ made by the water surface in the capillary with the wall.
A ferry boat has internal volume 1 m3 and weight 50 kg.(a) Neglecting the thickness of the wood, find the fraction of the volume of the boat immersed in water.(b) If a leak develops in the bottom and water starts coming in, what fraction of the boat's volume will be filled with water before water starts coming in from the sides?
A cube of ice floats partly in water and partly in K.oil (in the following figure). Find the ratio of the volume of ice immersed in water to that in K.oil. Specific gravity of K.oil is 0.8 and that of ice is 0.9.

A cubical metal block of edge 12 cm floats in mercury with one fifth of the height inside the mercury. Water in it. Find the height of the water column to be poured.
Specific gravity of mercury = 13.6.
How much amount of work is done in forming a soap bubble of radius r?
Explain the capillary action.
Derive an expression for capillary rise for a liquid having a concave meniscus.
The property of _______ of a liquid surface enables the water droplets to move upward in plants.
A certain number of spherical drops of a liquid of radius R coalesce to form a single drop of radius R and volume V. If T is the surface tension of the liquid, then
Why coffee runs up into a sugar lump (a small cube of sugar) when one corner of the sugar lump is held in the liquid?
A large number of liquid drops each of radius 'r' coalesce to form a big drop of radius 'R'. The energy released in the process in converted into kinetic energy of the big drop. The speed of the big drop is ______. (T = surface tension of liquid, p = density of liquid)
Two mercury droplets of radii 0.1 cm. and 0.2 cm. collapse into one single drop. What amount of energy is released? The surface tension of mercury T = 435.5 × 10–3 Nm–1.
A liquid flows out drop by drop from a vessel through a vertical tube with an internal diameter of 2 mm, then the total number of drops that flows out during 10 grams of the liquid flow out ______. [Assume that the diameter of the neck of a drop at the moment it breaks away is equal to the internal diameter of tube and surface tension is 0.02 N/m].
The surface tension of a soap solution is T. The work done in blowing a soap bubble of diameter d to that of a diameter 2d is ______.
