Advertisements
Advertisements
प्रश्न
State Einstein’s photoelectric equation. Explain all characteristics of the photoelectric effect, on the basis of Einstein’s photoelectric equation.
Advertisements
उत्तर
Einstein’s photoelectric equation: K.E.max = (hν – `phi_0`)
Characteristics of photoelectric effect:
- The photoelectric work function `phi_0` is constant for a given emitter. Hence if the frequency ‘ν’ of the incident radiation is decreased, the maximum kinetic energy of the emitted photoelectrons decreases, till it becomes zero for a certain frequency ν0. Therefore, from Einstein’s equation,
0 = hv0 - `phi_0`
∴ `phi_0` = hv0 .......….(1)
This shows that the threshold frequency is related to the work function of the metal and hence it has different values for different metals. - Using equation (1), Einstein’s equation can be written as
`1/2"mv"_"max"^2 = "hv" - "hv"_0`
∴ `1/2"mv"_"max"^2 = "h"("v" - "v"_0)`
This equation shows that:
a. If ν < ν0, then K.E is negative, which is not possible. In this case, photoelectric emission is not possible.
b. If ν > ν0, then photoelectrons move with some velocity.
∴ K.E > 0, which is possible. Hence, photoelectrons are emitted.
c. If ν = ν0, the photoelectrons are just emitted. In this case, K.E = 0. - The photoelectric equation is,
`1/2"mv"_"max"^2 = "hv" - phi_0` .....….(2)
where, hν = energy of the photon of incident radiation.
`phi_0 = "hv"_0` = photoelectric work function of the metal.
Thus, both the terms on the R.H.S of equation (2) depends on the frequency and not on the intensity of radiation. Hence the maximum kinetic energy with which photoelectrons are emitted is independent of the intensity of radiation. However, since `phi_0` and h are constants, the maximum kinetic energy of the photoelectrons is directly proportional to the frequency. - According to the quantum theory, when the intensity of the radiation increases, there is a proportional increase in the number of photons incident per second on the surface. One photon can cause the emission of one photoelectron. Therefore, with the increase in the intensity of radiation, there will be an increase in the photoelectron interactions and the rate of emission of electrons.
- The emission of a photoelectron is the result of a collision between a photon and an electron. As soon as the radiation is incident on the photosensitive surface, the entire energy of the photon is absorbed by the electron at once. Therefore, the electrons are emitted at a moment when light is incident on the metal surface. This explains why photoelectric emission is instantaneous.
- If the electron, with which the incident photon collides, is situated on the emitting surface, the electron will be ejected with maximum K.E as given by Einstein’s equation.
- If, however, the electron is situated in the interior of the emitting material, it will lose some energy in coming to the surface. This explains why the photoelectrons are emitted with different kinetic energies.
Thus, all the features of the photoelectric effect are explained.
APPEARS IN
संबंधित प्रश्न
Photocurrent recorded in the microammeter in an experimental setup of the photoelectric effect vanishes when the retarding potential is more than 0.8 V if the wavelength of incident radiation is 4950 Å. If the source of incident radiation is changed, the stopping potential turns out to be 1.2 V. Find the work function of the cathode material and the wavelength of the second source.
The threshold wavelength of tungsten is 2.76 x 10-5 cm.
(a) Explain why no photoelectrons are emitted when the wavelength is more than 2.76 x 10-5 cm.
(b) What will be the maximum kinetic energy of electrons ejected in each of the following cases
(i) if ultraviolet radiation of wavelength λ = 1.80 × 10-5 cm and
(ii) radiation of frequency 4 x 1015 Hz is made incident on the tungsten surface?
Radiation of wavelength 4500 Å is incident on a metal having work function 2.0 eV. Due to the presence of a magnetic field B, the most energetic photoelectrons emitted in a direction perpendicular to the field move along a circular path of radius 20 cm. What is the value of the magnetic field B?
Given the following data for incident wavelength and the stopping potential obtained from an experiment on the photoelectric effect, estimate the value of Planck's constant and the work function of the cathode material. What is the threshold frequency and corresponding wavelength? What is the most likely metal used for emitter?
| Incident wavelength (in Å) | 2536 | 3650 |
| Stopping potential (in V) |
1.95 | 0.5 |
The electrons are emitted in the photoelectric effect from a metal surface.
Find the energy of photon which have momentum 2 × 10-16 gm-cm/sec.
If the total energy of radiation of frequency 1014 Hz is 6.63 J, Calculate the number of photons in the radiation.
With the help of a circuit diagram describe the experiment to study the characteristics of the photoelectric effect. Hence discuss any 2 characteristics of the photoelectric effect.
The maximum velocity of the photoelectron emitted by the metal surface is 'v '. Charge and mass of the photoelectron is denoted by 'e' and 'm' respectively. The stopping potential in volt is ______.
The work function of a metallic surface is 5.01 eV. The photoelectrons are emitted when light of wavelength 2000 Å falls on it. The potential difference applied to stop the fastest photoelectrons is [h = 4.14 x 10-15 eV sec] ____________.
Photoelectrons emitted from a metallic surface are initially ____________.
When a surface 1 cm thick is illuminated by light of wavelength 'λ', the stopping potential is 'V0'. When the same surface is illuminated by light of wavelength '3λ', the stopping potential is `"V"_0/6`. The threshold wavelength for the metallic surface is ______.
Which one of the following statements ts INCORRECT for stopping potential in photoelectric emission?
A light of wavelength '`lambda`' and intensity 'I' falls on photosensitive material. If 'N' photoelectrons are emitted, each with kinetic energy E, then ____________.
A metal surface having work function 'w0' emits photoelectrons when photons of energy 'E' are incident on it. The electron enters the uniform magnetic field (B) in perpendicular direction and moves in circular path of radius 'r'. Then 'r' is equal to (m and e be the mass and charge of electron respectively) ____________.
The ratio of slopes m1: ro2 of the lines given in the following graphs is, ______.
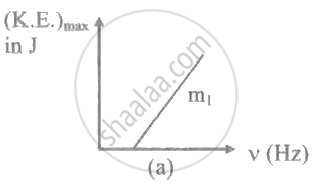
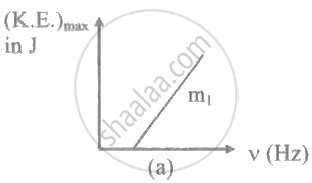
When radiation of wavelength λ is used to illuminate a metallic surface, the stopping potential is V. When the same surface is illuminated with radiation of wavelength 3λ, the stopping potential is `"V"/4`. If the threshold wavelength for the metallic surface is nλ. then value of n will be ______.
The radiation emitted, when an electron jumps from n = 3 to n = 2 orbit is a hydrogen atom, falls on a metal to produce photoelectron. The electrons from the metal surface with maximum kinetic energy are made to move perpendicular to a magnetic field of `1/320`T in a radius of 10-3m. Find the 320 work function of metal:
When ultraviolet light of wavelength 100 nm is incident upon a sample of silver metal, a potential difference of 7.7 volt is required to stop the photoelectrons from reaching the collector plate. The potential required to stop photo electrons when light of wavelength 200 nm is incident upon silver is ______.
The maximum kinetic energy of the photoelectrons ejected will be ______ eV when the light of wavelength 350 nm is incident on a cesium surface. The work function of cesium = 1.9 eV.
We wish to observe an object which is 2.5Å in size. The minimum energy photon that can be used ______.
For a given photosensitive material and frequency (> threshold frequency) of incident radiation, the photoelectric current varies with the intensity of incident light as:
Give Einstein's explanation of the photoelectric effect.
A parallel plate capacitor with air between the plates has capacitance 9 pF. The separation between the plates becomes thrice and the space between them is filled with a medium of dielectric constant 6. The capacitance becomes 'x' pF. The value of 'x' is ______.
According to Einstein's photoelectric equation, the graph of kinetic energy of the photoelectron emitted from the metal versus the frequency of incident radiation gives a straight line graph whose slopе ______.
