Advertisements
Advertisements
प्रश्न
State Einstein’s photoelectric equation. Explain all characteristics of the photoelectric effect, on the basis of Einstein’s photoelectric equation.
Advertisements
उत्तर
Einstein’s photoelectric equation: K.E.max = (hν – `phi_0`)
Characteristics of photoelectric effect:
- The photoelectric work function `phi_0` is constant for a given emitter. Hence if the frequency ‘ν’ of the incident radiation is decreased, the maximum kinetic energy of the emitted photoelectrons decreases, till it becomes zero for a certain frequency ν0. Therefore, from Einstein’s equation,
0 = hv0 - `phi_0`
∴ `phi_0` = hv0 .......….(1)
This shows that the threshold frequency is related to the work function of the metal and hence it has different values for different metals. - Using equation (1), Einstein’s equation can be written as
`1/2"mv"_"max"^2 = "hv" - "hv"_0`
∴ `1/2"mv"_"max"^2 = "h"("v" - "v"_0)`
This equation shows that:
a. If ν < ν0, then K.E is negative, which is not possible. In this case, photoelectric emission is not possible.
b. If ν > ν0, then photoelectrons move with some velocity.
∴ K.E > 0, which is possible. Hence, photoelectrons are emitted.
c. If ν = ν0, the photoelectrons are just emitted. In this case, K.E = 0. - The photoelectric equation is,
`1/2"mv"_"max"^2 = "hv" - phi_0` .....….(2)
where, hν = energy of the photon of incident radiation.
`phi_0 = "hv"_0` = photoelectric work function of the metal.
Thus, both the terms on the R.H.S of equation (2) depends on the frequency and not on the intensity of radiation. Hence the maximum kinetic energy with which photoelectrons are emitted is independent of the intensity of radiation. However, since `phi_0` and h are constants, the maximum kinetic energy of the photoelectrons is directly proportional to the frequency. - According to the quantum theory, when the intensity of the radiation increases, there is a proportional increase in the number of photons incident per second on the surface. One photon can cause the emission of one photoelectron. Therefore, with the increase in the intensity of radiation, there will be an increase in the photoelectron interactions and the rate of emission of electrons.
- The emission of a photoelectron is the result of a collision between a photon and an electron. As soon as the radiation is incident on the photosensitive surface, the entire energy of the photon is absorbed by the electron at once. Therefore, the electrons are emitted at a moment when light is incident on the metal surface. This explains why photoelectric emission is instantaneous.
- If the electron, with which the incident photon collides, is situated on the emitting surface, the electron will be ejected with maximum K.E as given by Einstein’s equation.
- If, however, the electron is situated in the interior of the emitting material, it will lose some energy in coming to the surface. This explains why the photoelectrons are emitted with different kinetic energies.
Thus, all the features of the photoelectric effect are explained.
APPEARS IN
संबंधित प्रश्न
Using the values of work function given in the following table, tell which metal will require the highest frequency of incident radiation to generate photocurrent.
Typical values of work function for some common metals
| Metal | Work function (in eV) |
| Potassium | 2.3 |
| Sodium | 2.4 |
| Calcium | 2.9 |
| Zinc | 3.6 |
| Silver | 4.3 |
| Aluminium | 4.3 |
| Tungsten | 4.5 |
| Copper | 4.7 |
| Nickel | 5.0 |
| Gold | 5.1 |
It is observed in an experiment on the photoelectric effect that an increase in the intensity of the incident radiation does not change the maximum kinetic energy of the electrons. Where does the extra energy of the incident radiation go? Is it lost? State your answer with explanatory reasoning.
As the intensity of incident light increases ______
The minimum frequency for photoelectric effect on metal is 7 × 1014 Hz, Find the work function of the metal.
What is the photoelectric effect? Define stopping potential and photoelectric work function.
The energy of a photon is 2 eV. Find its frequency and wavelength.
The maximum velocity of photoelectron emitted is 4.8 m/s. If the e/m ratio of the electron is 1.76 × 1011 C/kg, then stopping potential is given by ______
The ratio of energies of photons produced due to transition of electron of hydrogen atom from its (i) second to first energy level and (ii) highest energy level to second level is respectively.
The kinetic energy of the most energetic photoelectron emitted from a metal surface is doubled when the wavelength of the incident radiation is reduced from λ1 to λ2. The work function of the metal is ______
The following graph shows the stopping potential V0 versus frequency v for photoelectric emission from two metals A and B. The slope of each of the lines gives ______

When a photon enters glass from air, which one of the following quantity does not change?
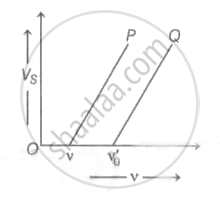
The graph of stopping potential `"V"_"s"` against frequency v of incident radiation is plotted for two different metals P and Q as shown in the graph. ΦP and ΦQ are work-functions of P and Q respectively, then
Light of different frequencies, whose photons have energies 3 eV and 18 eV respectively, successively illuminate a metal of work function 2 eV. The ratio of the maximum speeds of the emitted electrons will be ______.
The lowest frequency of light that will cause the emission of photoelectrons from the surface of a metal (for which work function is 1.65 eV) will be ____________.
The photon of frequency vis incident on a metal surface whose threshold frequency is v0. The kinetic energy of the emitted photoelectrons will be ______.
The radiations of energies 1 eV and 2.5 eV are incident on a metal surface having work function 0.5 eV. The ratio of the maximum velocities of the emitted photo-electrons is ____________.
When a certain metallic surface is illuminated with monochromatic light of wavelength '`lambda`', the stopping potential for photoelectric effect is '3V0'. If the same surface is illuminated with a light of wavelength '`2 lambda`', the stopping potential is found as 'V0'. The threshold wavelength for this surface is ____________.
When ultraviolet light of wavelength 100 nm is incident upon a sample of silver metal, a potential difference of 7.7 volt is required to stop the photoelectrons from reaching the collector plate. The potential required to stop photo electrons when light of wavelength 200 nm is incident upon silver is ______.
The maximum kinetic energy of the photoelectrons ejected will be ______ eV when the light of wavelength 350 nm is incident on a cesium surface. The work function of cesium = 1.9 eV.
A point isotropic light source of power P = 12 watts is located on the axis of a circular mirror of radius R = 3 cm. If the distance of the source from the centre of the mirror is a = 39 cm and the reflection coefficient of the mirror is α = 0.70 then the force exerted by the light ray on the mirror is ______ × 10-10 N.
If the electron in hydrogen atom jumps from second Bohr orbit to ground state and difference between energies of the two states is radiated in the form of photons. If the work function of the material is 4.2 eV, then stopping potential is ______.
[Energy of electron in nth orbit = `-13.6/"n"^2` eV ]
On a photosensitive material when frequency of incident radiation is increased by 30%, kinetic energy of emitted photoelectrons increases from 0.4 eV. The work function of the surface is ______.
Light of wavelength λ, which is less than threshold wavelength is incident on a photosensitive material. If incident wavelength is decreased so that emitted photoelectrons are moving with same velocity, then stopping potential will ______.
The following graphs show the variation of stopping potential corresponding to the frequency of incident radiation (ν) for a given metal. The correct variation is shown in graph [ν0 = threshold frequency].
|
(A) |
(B) |
|
(C) |
(D) |
Photoelectric emission is observed from a metallic surface for frequencies ν1 and ν2 of the incident light rays (ν1 > ν2). If the ratio of the maximum value of the kinetic energy of the photoelectrons emitted in the first case to that in the second case is 2 : K, then the threshold frequency of the metallic surface is ______.
Explain the failure of wave theory of light to account for the observations from experiments on photoelectric effect.
Explain the formation of clouds at high altitude.
In a photoelectric experiment, the stopping potential is 1.5V. What is the maximum kinetic energy of a photoelectron?
According to Einstein's photoelectric equation, the graph of kinetic energy of the photoelectron emitted from the metal versus the frequency of incident radiation gives a straight line graph whose slopе ______.




