Advertisements
Advertisements
प्रश्न
With the help of a circuit diagram describe the experiment to study the characteristics of the photoelectric effect. Hence discuss any 2 characteristics of the photoelectric effect.
With a neatly labelled circuit diagram, describe an experiment to study the characteristics of the photoelectric effect.
Advertisements
उत्तर
- A laboratory experimental set-up for the photoelectric effect consists of an evacuated glass tube with a quartz window.
- The glass tube contains photosensitive metal plates. One is the emitter E and another plate is the collector C.

Schematic of experimental set-up for the photoelectric effect - The emitter and collector are connected to a voltage source whose voltage can be changed and to an ammeter to measure the current in the circuit.
- A potential difference of V, as measured by the voltmeter, is maintained between the emitter E and collector C. Generally, C (the anode) is at a positive potential with respect to the emitter E (the cathode). This potential difference can be varied and C can even be at a negative potential with respect to E.
- When the anode potential (V) is positive, it accelerates the electrons. This potential is called accelerating potential. When the anode potential (V) is negative, it retards the flow of electrons. This potential is known as retarding potential.
- A source S of monochromatic light of sufficiently high frequency (short wavelength ≤ 10–7 m) is used.
Two characteristics of the photoelectric effect:
- The photoelectric work function `phi_0` is constant for a given emitter. Hence if the frequency ‘ν’ of the incident radiation is decreased, the maximum kinetic energy of the emitted photoelectrons decreases, till it becomes zero for a certain frequency ν0.
Therefore, from Einstein’s equation,
0 = `"hv"_0 - phi_0`
∴ `phi_0 = "hv"_0` ........(1)
This shows that the threshold frequency is related to the work function of the metal and hence it has different values for different metals. - The photoelectric equation is,
`1/2"mv"_"max"^2 = "hv" - phi_0` ........(2)
where, hν = energy of the photon of incident radiation.
`phi_0 = "hv"_0` = photoelectric work function of the metal.
Thus, both the terms on the R.H.S of equation (2) depend on the frequency and not on the intensity of radiation. Hence the maximum kinetic energy with which photoelectrons are emitted is independent of the intensity of radiation. However, since `phi_0` and h are constants, the maximum kinetic energy of the photoelectrons is directly proportional to the frequency.
संबंधित प्रश्न
Can microwaves be used in the experiment on photoelectric effect?
Using the values of work function given in the following table, tell which metal will require the highest frequency of incident radiation to generate photocurrent.
Typical values of work function for some common metals
| Metal | Work function (in eV) |
| Potassium | 2.3 |
| Sodium | 2.4 |
| Calcium | 2.9 |
| Zinc | 3.6 |
| Silver | 4.3 |
| Aluminium | 4.3 |
| Tungsten | 4.5 |
| Copper | 4.7 |
| Nickel | 5.0 |
| Gold | 5.1 |
It is observed in an experiment on the photoelectric effect that an increase in the intensity of the incident radiation does not change the maximum kinetic energy of the electrons. Where does the extra energy of the incident radiation go? Is it lost? State your answer with explanatory reasoning.
As the intensity of incident light increases ______
Draw a neat labelled diagram of a schematic of the experimental setup for the photoelectric effect.
Explain the concept of the photoelectric effect.
Which one of the following is TRUE in photoelectric emission?
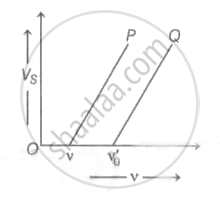
The graph of stopping potential `"V"_"s"` against frequency v of incident radiation is plotted for two different metals P and Q as shown in the graph. ΦP and ΦQ are work-functions of P and Q respectively, then
For photoelectric emission from certain metal, the cut-off frequency is v. If radiation of frequency 2v impinges on the metal plate, the maximum possible velocity of the emitted electron will be (m is the electron mass) ____________.
In photoelectric experiment, if both the intensity and frequency of the incident light are doubled, then the saturation of photoelectric current ______.
Light of wavelength `lambda` strikes a photo-sensitive surface and electrons are ejected with kinetic energy E. If the kinetic energy is to be increased to 2E, the wavelength must be changed to `lambda'` where ____________.
When certain metal surface is illuminated with a light of wavelength A., the stopping potential is V, When the same surface is illuminated by light of wavelength 2λ, the stopping potential is `("V"/3)`. The threshold wavelength for the surface is ______.
When wavelength of incident radiation on the metal surface is reduced from 'λ1' to 'λ2', the kinetic energy of emitted photoelectrons is tripled. The work function of the metal is ______.
(h = Planck's constant, c =velocity of light)
The photo electric effect to take place for a metal, the minimum frequency required is 5.792 × 1014 Hz. A light of wavelength 6000 Å is incident on that metal surface. What is the corresponding frequency of light and will there be photoelectric emissions? [velocity of light = 3 × 108 m/s]
Light of different frequencies, whose photons have energies 3 eV and 18 eV respectively, successively illuminate a metal of work function 2 eV. The ratio of the maximum speeds of the emitted electrons will be ______.
The photon of frequency vis incident on a metal surface whose threshold frequency is v0. The kinetic energy of the emitted photoelectrons will be ____________.
The lowest frequency of light that will cause the emission of photoelectrons from the surface of a metal (for which work function is 1.65 eV) will be ____________.
The photon of frequency vis incident on a metal surface whose threshold frequency is v0. The kinetic energy of the emitted photoelectrons will be ______.
Which one of the following statements ts INCORRECT for stopping potential in photoelectric emission?
The radiations of energies 1 eV and 2.5 eV are incident on a metal surface having work function 0.5 eV. The ratio of the maximum velocities of the emitted photo-electrons is ____________.
The stopping potential in the context of photoelectric effect depends on the following property of incident electromagnetic radiation ______.
When radiation of wavelength λ is used to illuminate a metallic surface, the stopping potential is V. When the same surface is illuminated with radiation of wavelength 3λ, the stopping potential is `"V"/4`. If the threshold wavelength for the metallic surface is nλ. then value of n will be ______.
The radiation emitted, when an electron jumps from n = 3 to n = 2 orbit is a hydrogen atom, falls on a metal to produce photoelectron. The electrons from the metal surface with maximum kinetic energy are made to move perpendicular to a magnetic field of `1/320`T in a radius of 10-3m. Find the 320 work function of metal:
The photoelectric threshold for a certain metal surface is 3600 Å. If the metal surface is irradiated by a wavelength of 1100 Å, then kinetic energy of the emitted photoelectrons is ______.
If the maximum kinetic energy of emitted electrons in the photoelectric effect is 2eV, the stopping potential will be ______.
Explain the failure of wave theory of light to account for the observations from experiments on photoelectric effect.
Light of wavelength 'λ' falls on a metal having work function \[\frac {hc}{λ_0}\]. Photoelectric effect will take place only if (λ0 is the threshold wavelength) ______.
A parallel plate capacitor with air between the plates has capacitance 9 pF. The separation between the plates becomes thrice and the space between them is filled with a medium of dielectric constant 6. The capacitance becomes 'x' pF. The value of 'x' is ______.
