Advertisements
Advertisements
प्रश्न
Reaction of \[\ce{C6H5CH2Br}\] with aqueous sodium hydroxide follows ______.
विकल्प
SN1 mechanism
SN2 mechanism
Any of the above two depending upon the temperature of reaction
Saytzeff rule
Advertisements
उत्तर
Reaction of \[\ce{C6H5CH2Br}\] with aqueous sodium hydroxide follows SN1 mechanism.
Explanation:
Greater the stability of carbonation, greater will be its ease of formation from alkyl halide and faster will be the rate of reaction. In case of alkyl halides, 3° carbocations.
For the same reasons, allylic and benzylic halides show high reactivity towards the SN1 reaction. The carbonation thus formed gets stabilized through resonance as shown below:
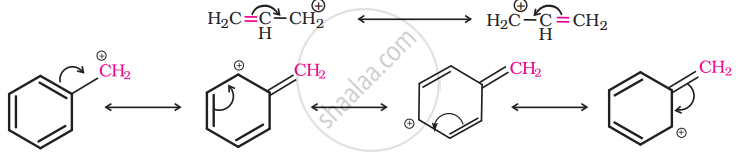
So, as the given compound, \[\ce{C6HCH2Cl}\] is a benzylic halide, it would undergo SN1 reaction.
APPEARS IN
संबंधित प्रश्न
Give reasons : n-Butyl bromide has higher boiling point than t-butyl bromide.
Arrange the set of compounds in order of increasing boiling points.
Bromomethane, Bromoform, Chloromethane, Dibromomethane.
Explain why alkyl halides, though polar, are immiscible with water?
p-Dichlorobenzene has higher m.p. and lower solubility than those of o- and m-isomers. Discuss.
Why dextro and laevorotatory isomers of Butan-2-ol are difficult to separate by fractional distillation?
For the same alkyl group, an alkyl bromide has a higher boiling point than alkyl fluoride because:
Which of the following compounds has the highest boiling point?
Which of the following possesses the highest melting point?
p-dichlorobenzene has a higher melting point than its o- and m-isomers because ____________.
Out of o-and p-dibromobenzene which one has higher melting point and why?
Arrange the following compounds in increasing order of their boiling points:
CH3CH2OH, CH3−CHO, CH3−COOH
Why alkyl halides though polar are immiscible with water?
Why is the boiling point of o-dichlorobenzene higher than p-dichlorobenzene, but the melting point of para-isomer is higher than ortho-isomer?
Arrange the isomeric dichlorobenzene in the increasing order of their boiling point and melting points.
Write the structure of the following organic halogen compound:
4-tert-Butyl-3-iodoheptane
Name the following halides according to the IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
\[\ce{CH3 C(C2H5)2CH2Br}\]
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
