Advertisements
Advertisements
प्रश्न
Arrange the following compounds in increasing order of their boiling points:
CH3CH2OH, CH3−CHO, CH3−COOH
Advertisements
उत्तर
The increasing order of boiling point in the following compounds is CH3−CHO < CH3CH2OH < CH3−COOH. Ethanoic acid and ethanol have a comparatively higher boiling point than ethanal, as they both are held by strong hydrogen bonds in between them, whereas in ethanal there is a dipole-dipole interaction between them.
APPEARS IN
संबंधित प्रश्न
Give reasons : n-Butyl bromide has higher boiling point than t-butyl bromide.
Arrange the set of compounds in order of increasing boiling points.
Bromomethane, Bromoform, Chloromethane, Dibromomethane.
p-Dichlorobenzene has higher m.p. and lower solubility than those of o- and m-isomers. Discuss.
Which of the following compounds has the highest boiling point?
Which of the following possesses the highest melting point?
Arrange the following compounds in the increasing order of their densities.
(a)

(b)

(c)
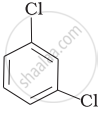
(d)

Reaction of \[\ce{C6H5CH2Br}\] with aqueous sodium hydroxide follows ______.
Arrange the isomeric dichlorobenzene in the increasing order of their boiling point and melting points.
Write the structure of the following organic halogen compound.
1,4-Dibromobut-2-ene
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
