Advertisements
Advertisements
प्रश्न
Out of (CH3)3 C-Br and (CH3)3 C-I, which one is more reactive towards SN1 and why?
Advertisements
उत्तर
In SN1 reactions, reactivity depends on the stability of carbocation after removing the leaving group form the reactant. Since the carbocation is the same here, so, we will see the tendency of leaving the group. As we can see in the following reactions Br and r are the leaving groups, out of them r is a better leaving group. Hence SN1 reaction will be faster in (CH3)3 C-I (CH3)3 - Br
\[\begin{array}{cc}
\ce{CH3}\phantom{..............}\ce{CH3}\\
\phantom{.}|\phantom{.................}|\phantom{...}\\
\ce{CH3-C-Br->CH3-C^+ + Br^-}\\
\phantom{.}|\phantom{.................}|\phantom{...}\\\ce{CH3}\phantom{..............}\ce{CH3}\\
\end{array}\]
\[\begin{array}{cc}
\phantom{...}\ce{CH3}\phantom{............}\ce{CH3}\\
\phantom{...}|\phantom{...............}|\phantom{...}\\
\ce{CH3-C-I->CH3-C^+ + I^-}\\
\phantom{...}|\phantom{...............}|\phantom{...}\\
\phantom{...}\ce{CH3}\phantom{............}\ce{CH3}\\
\end{array}\]
संबंधित प्रश्न
How the following conversion can be carried out?
Chlorobenzene to p-nitrophenol
Give reasons:
The dipole moment of chlorobenzene is lower than that of cyclohexyl chloride.
Which of the following compounds will give racemic mixture on nucleophilic substitution by \[\ce{OH-}\] ion?
(a) \[\begin{array}{cc}
\phantom{}\ce{CH3 - CH - Br}\\
\phantom{}|\\
\phantom{....}\ce{C2H5}\phantom{}
\end{array}\]
(b) \[\begin{array}{cc}
\phantom{..}\ce{Br}\\
\phantom{}|\\
\phantom{}\ce{CH3 - C - CH3}\\
\phantom{}|\\
\phantom{....}\ce{C2H5}\phantom{}
\end{array}\]
(c) \[\begin{array}{cc}
\phantom{....}\ce{CH3 - CH - CH2Br}\\
\phantom{}|\\
\phantom{....}\ce{C2H5}\phantom{}
\end{array}\]
Arrange the following compounds in increasing order of rate of reaction towards nucleophilic substitution.
| (a) |  |
| (b) | 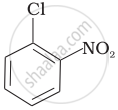 |
| (c) |  |
Arrange the following compounds in increasing order of rate of reaction towards nucleophilic substitution.
| (a) |  |
| (b) |  |
| (c) |  |
Arrange the following compounds in increasing order of rate of reaction towards nucleophilic substitution.
| (a) | 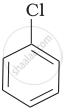 |
| (b) | 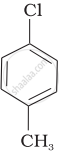 |
| (c) |  |
\[\ce{C6H12O6 ->[(Zymase)] A ->[NaOH][\Delta] B + CHI3}\]
The number of carbon atoms present in the product B is:
Why haloarenes are not reactive towards nucleophilic substitution reaction? Give two reactions.
In the reaction, \[\mathrm{CH}_3\mathrm{C}\equiv\mathrm{\overline{C}Na}^++(\mathrm{CH}_3)_2\mathrm{CHCl}\to\] the product formed is ______.
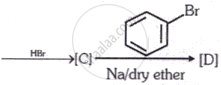
Identify the final product [D] obtained in the following sequence of reactions.
\[\ce{CH3CHO \underset{ii) H2O+}{\overset{i) LiAlH4}{->}} [A] \underset{\triangle}{\overset{H2SO4}{->}} [B]}\]