Advertisements
Advertisements
प्रश्न
Write chemical equation in support of your answer.
Out of  Cl and
Cl and 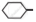 CH2- Cl, which one is more reactive towards nucleophilic substitution reaction and why?
CH2- Cl, which one is more reactive towards nucleophilic substitution reaction and why?
Advertisements
उत्तर
 CH2 - Cl , will be more reactive towards SN2 reaction as in case of reaction as in case of
CH2 - Cl , will be more reactive towards SN2 reaction as in case of reaction as in case of  Cl the back side attack will be very difficult.
Cl the back side attack will be very difficult.
APPEARS IN
संबंधित प्रश्न
Chlorobenzene is extremely less reactive towards a nucleophilic substitution reaction. Give two reasons for the same.
The presence of nitro group (−NO2) at o/p positions increases the reactivity of haloarenes towards nucleophilic substitution reactions.
How the following conversion can be carried out?
Chlorobenzene to p-nitrophenol
Give reasons:
The dipole moment of chlorobenzene is lower than that of cyclohexyl chloride.
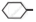
Arrange the following compounds in increasing order of rate of reaction towards nucleophilic substitution.
| (a) | 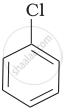 |
| (b) | 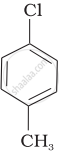 |
| (c) |  |
Allyl chloride is hydrolysed more readily than n-propyl chloride. Why?
Assertion: Chlorobenzene is resistant to nucleophilic substitution reaction at room temperature.
Reason (R): C–Cl bond gets weaker due, to resonance.
Why haloarenes are not reactive towards nucleophilic substitution reaction? Give two reactions.
In the reaction, \[\mathrm{CH}_3\mathrm{C}\equiv\mathrm{\overline{C}Na}^++(\mathrm{CH}_3)_2\mathrm{CHCl}\to\] the product formed is ______.
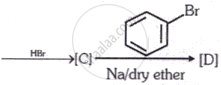
Identify the final product [D] obtained in the following sequence of reactions.
\[\ce{CH3CHO \underset{ii) H2O+}{\overset{i) LiAlH4}{->}} [A] \underset{\triangle}{\overset{H2SO4}{->}} [B]}\]