Advertisements
Advertisements
प्रश्न
Explain, why a gas bubble released at the bottom of a lake grows in size as it rises to the surface of the lake.
टिप्पणी लिखिए
Advertisements
उत्तर
Lake has greater pressure at the bottom than the surface as pressure increases with depth. So when the gas bubble is released at the bottom of the lake it experiences more pressure and is small in size but as it rises upwards the pressure experienced by it decreases. So it grows in size as it moves towards the surface from bottom.
shaalaa.com
Variation of Pressure with Depth in a Fluid
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
APPEARS IN
संबंधित प्रश्न
Calculate the pressure exerted by 0.8 m vertical length of alcohol of density 0.80 gcnr5 in SI units.
[Take g = 10 ms−2].
What is the pressure exerted by 75 cm vertical column of mercury of density 13600 kgm−3 in SI units?
[Take g = 9.8 ms−2].
Calculate the equivalent height of mercury, which will exert as much pressure as 960 m of seawater of density 1040 kgm−3. The density of mercury is 13600 kgm−3.
The pressure of water on the ground floor, in a water pipe is 150000 Pa, whereas pressure on the fourth floor is 30000 Pa. Calculate the height of the fourth floor. Take g = 10 ms−2.
What do you mean by pressure? Write its SI unit.
Is pressure a scalar or vector physical quantity?
State whether thrust is scalar or vector.
What fact about liquid pressure does the following diagram in the following figure illustrate?
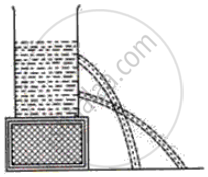
What is a manometer? How does it show whether the pressure inside a vessel connected to one arm of it, is lower or above the atmospheric pressure?
What is the SI unit of density?
