Advertisements
Advertisements
प्रश्न
Advertisements
उत्तर
APPEARS IN
संबंधित प्रश्न
Calculate the pressure exerted by 0.8 m vertical length of alcohol of density 0.80 gcnr5 in SI units.
[Take g = 10 ms−2].
What vertical height of water will exert a pressure of 333200 Pa? The density of water is 1000 kgnr3 and g = 9.8 ms−2.
Calculate the equivalent height of mercury, which will exert as much pressure as 960 m of seawater of density 1040 kgm−3. The density of mercury is 13600 kgm−3.
The pressure of water on the ground floor is 160000 Pa. Calculate the pressure on the fifth floor, at a height of 15 m.
The normal pressure of air is 76 cm of mercury. Calculate the pressure in SI units.
[Density of mercury = 13600 kg/m3 and g = 10 m/s2]
What fact about liquid pressure does the following diagram in the following figure illustrate?
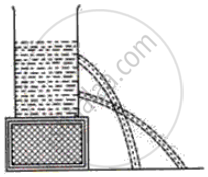
What is the SI unit of density?
