Advertisements
Advertisements
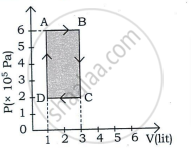
प्रश्न
Draw a p-V diagram showing positive work with varying pressure.
Advertisements
उत्तर
p-V diagram showing positive work with varying pressure:

APPEARS IN
संबंधित प्रश्न
For work done to be reversible, the process should be ______
Draw a p-V diagram showing negative work with varying pressure.
Differentiate between the reversible and irreversible processes.
Explain graphically (i) positive work with varying pressure, (ii) negative work with varying pressure, and (iii) positive work at constant pressure.
Explain the thermodynamics of the isobaric process.
When you exercise in the morning, by considering your body as a thermodynamic system, which of the following is true?
In an isochoric process, we have ____________.
Apply first law for an adiabatic process.
Apply first law for an isobaric process.
Draw the PV diagram for the adiabatic process.
Explain the isobaric process and derive the work done in this process.
What are the limitations of the first law of thermodynamics?
Draw the TP diagram (P-x axis, T-y axis), VT(T-x axis, V-y axis) diagram for
- Isochoric process
- Isothermal process
- Isobaric process
An ideal gas is taken in a cyclic process as shown in the figure. Calculate
- work done by the gas
- work done on the gas
- Net work done in the process

A thermodynamic system undergoes cyclic process ABCDA as shown in the figure. The work done by the system is ______

A monoatomic gas of pressure p having volume V expands isothermally to a volume 2V and then adiabatically to a volume 16V. The final pressure of the gas is ____________.
`("ratio of specific heats" = 5/3)`
Among the amount of heat absorbed and the amount of work done by a system, ______
An ideal gas is made to go from a state A to stale B in the given two different ways (see figure) (i) an isobaric and then an isochoric process and (ii) an isochoric and then an isobaric process. The work done by gas in the two processes are W1 and W2 respectively. Then,

One mole of an ideal gas with `gamma` = 1.4 is adiabatically compressed so that its temperature rises from 27° C to 47° C. The change in the internal energy of the gas is (R = 8.3 J/mol.K) ____________.
Two identical samples of a gas are allowed to expand (i) isothermally (ii) adiabatically. Work done is ____________.
For an isothermal expansion of a perfect gas, the value of `(Delta "P")/"P"` is equal to ____________.
In an isothermal process, the volume of an ideal gas is halved. One can say that ____________.
An ideal gas is compressed to half its initial volume by means of several processes. Which of the process results in the maximum work done on the gas?
In a certain thermodynamical process, the pressure of a gas depends on its volume as kV3. The work done when the temperature changes from 100°C to 300°C will be ______ nR, where n denotes number of moles of a gas.
When an inflated ballon is suddenly burst, why is the emerging air slightly cooled?
An ideal gas is taken through a cyclic process ABCDA as shown in figure. The net work done by the gas during the cycle is ______.
Explain how can a gas be expanded at constant temperature.
In a cyclic process, if ΔU = internal energy, W = work done, Q = Heat supplied then ______.
