Advertisements
Advertisements
प्रश्न
Cutoff wavelength of X-rays coming from a Coolidge tube depends on the
(a) target material
(b) accelerating voltage
(c) separation between the target and the filament
(d) temperature of the filament.
विकल्प
target material
accelerating voltage
separation between the target and the filament
temperature of the filament.
Advertisements
उत्तर
accelerating voltage
Cutoff wavelength (`lambda_min`) is given by `lambda_min = (hc)/(eV)`
Here,
h = Planck's constant
c = Speed of light
V = Accelerating voltage
e = Charge of electron
Clearly, a cutoff wavelength depends on accelerating voltage. It does not depend on the target material, separation between the target and the temperature of the filament.
APPEARS IN
संबंधित प्रश्न
Identify the part of the electromagnetic spectrum which is:
produced by bombarding a metal target by high speed electrons.
Use the formula λm T= 0.29 cm K to obtain the characteristic temperature ranges for different parts of the electromagnetic spectrum. What do the numbers that you obtain tell you?
Name the electromagnetic radiations used for (a) water purification, and (b) eye surgery.
What is the range of the wavelength of the following electromagnetic waves?
(a) Micro waves .
Which part of electromagnetic spectrum is used in radar systems?
Consider a photon of a continuous X-ray coming from a Coolidge tube. Its energy comes from
If the current in the circuit for heating the filament is increased, the cutoff wavelength
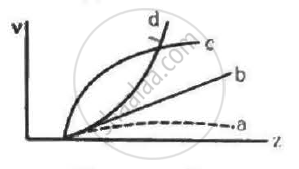
Frequencies of Kα X-rays of different materials are measured. Which one of the graphs in the figure may represent the relation between the frequency v and the atomic number Z ?
The X-ray beam emerging from an X-ray tube
For harder X-rays,
(a) the wavelength is higher
(b) the intensity is higher
(c) the frequency is higher
(d) the photon energy is higher.
The Kα X-rays of aluminium (Z = 13) and zinc (Z = 30) have wavelengths 887 pm and 146 pm respectively. Use Moseley's law √v = a(Z − b) to find the wavelengths of the Kα X-ray of iron (Z = 26).
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
The Kα and Kβ X-rays of molybdenum have wavelengths 0.71 A and 0.63 A respectively. Find the wavelength of Lα X-ray of molybdenum.
Name three radiations and their wavelength range which are invisible and beyond the violet end of the visible spectrum.
Name the radiation which can be detected by thermopile.
How will you investigate the existence of the radiation beyond the red and violet extremes of the spectrum?
State three properties of infrared radiations similar to that of visible light.
Answer briefly.
Give two uses of radio waves.
Electromagnetic waves of wavelengths λ1, λ2 and λ3 are used in a radar system, in water purifiers and in remote switches of TV, respectively.
- Identify the electromagnetic waves.
- Write one source for each of them.
Identify the electromagnetic wave whose wavelength range is from about 10-12 m to about 10-8 m. Write one use of this.
