Advertisements
Advertisements
प्रश्न
Consider a metal exposed to light of wavelength 600 nm. The maximum energy of the electron doubles when light of wavelength 400 nm is used. Find the work function in eV.
Advertisements
उत्तर
Work function (or threshold energy) (W0): The minimum energy of incident radiation required to eject the electrons from the metallic surface is defined as the work function of that surface.
W0 = hv0 = `(hc)/λ_0` Joules; v0 = Threshold frequency; λ0 = Threshold wavelength
Work function in electron volt, W0(eV) = `(hc)/(eλ_0) = 12375/(λ_0(Å))`
Einstein's photoelectric equation is E = W0 + Kmax
Maximum energy = `hv - phi`
According to the problem for the first condition wavelength of light λ = 600 nm and for the second condition, the wavelength of light λ' = 400 nm
Also, the maximum kinetic energy for the second condition is equal to twice the kinetic energy in the first condition.
i.e., K'max = 2Kmax
then K'max = `(hc)/λ - phi`
⇒ 2Kmax = `(hc)/λ^' - phi`
⇒ `2(1230/600 - phi) = (1230/400 - phi)` ......[∵ hc = 1240 eV nm]
⇒ `phi = 1230/1230` = 1.02 eV
APPEARS IN
संबंधित प्रश्न
Every metal has a definite work function. Why do all photoelectrons not come out with the same energy if incident radiation is monochromatic? Why is there an energy distribution of photoelectrons?
Can we find the mass of a photon by the definition p = mv?
The threshold wavelength of a metal is λ0. Light of wavelength slightly less than λ0 is incident on an insulated plate made of this metal. It is found that photoelectrons are emitted for some time and after that the emission stops. Explain.
Planck's constant has the same dimensions as
Let nr and nb be the number of photons emitted by a red bulb and a blue bulb, respectively, of equal power in a given time.
A photon of energy hv is absorbed by a free electron of a metal with work-function hv − φ.
In which of the following situations, the heavier of the two particles has smaller de Broglie wavelength? The two particles
(a) move with the same speed
(b) move with the same linear momentum
(c) move with the same kinetic energy
(d) have fallen through the same height
Calculate the momentum of a photon of light of wavelength 500 nm.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
Calculate the number of photons emitted per second by a 10 W sodium vapour lamp. Assume that 60% of the consumed energy is converted into light. Wavelength of sodium light = 590 nm
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
A parallel beam of monochromatic light of wavelength 663 nm is incident on a totally reflecting plane mirror. The angle of incidence is 60° and the number of photons striking the mirror per second is 1.0 × 1019. Calculate the force exerted by the light beam on the mirror.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
Find the maximum kinetic energy of the photoelectrons ejected when light of wavelength 350 nm is incident on a cesium surface. Work function of cesium = 1.9 eV
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
Explain how does (i) photoelectric current and (ii) kinetic energy of the photoelectrons emitted in a photocell vary if the frequency of incident radiation is doubled, but keeping the intensity same?
Show the graphical variation in the above two cases.
In photoelectric effect the photo current ______.
The work function for a metal surface is 4.14 eV. The threshold wavelength for this metal surface is ______.
The graph shows the variation of photocurrent for a photosensitive metal
- What does X and A on the horizontal axis represent?
- Draw this graph for three different values of frequencies of incident radiation ʋ1, ʋ2 and ʋ3 (ʋ3 > ʋ2 > ʋ1) for the same intensity.
- Draw this graph for three different values of intensities of incident radiation I1, I2 and I3 (I3 > I2 > I1) having the same frequency.
Read the following paragraph and answer the questions.
| The figure shows the variation of photoelectric current measured in a photocell circuit as a function of the potential difference between the plates of the photocell when light beams A, B, C and D of different wavelengths are incident on the photocell. Examine the given figure and answer the following questions: |

- Which light beam has the highest frequency and why?
- Which light beam has the longest wavelength and why?
- Which light beam ejects photoelectrons with maximum momentum and why?
Plot a graph showing the variation of photoelectric current, as a function of anode potential for two light beams having the same frequency but different intensities I1 and I2 (I1 > I2). Mention its important features.
What is the effect of threshold frequency and stopping potential on increasing the frequency of the incident beam of light? Justify your answer.
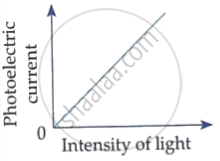
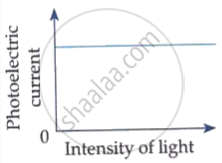
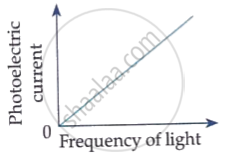
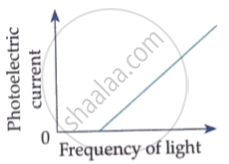
Which of the following options represents the variation of photoelectric current with property of light shown on the x-axis?