Advertisements
Advertisements
प्रश्न
Cement, the important building material is a mixture of oxides of several elements. Besides calcium, iron and sulphur, oxides of elements of which of the group (s) are present in the mixture?
विकल्प
Group 2
Groups 2, 13 and 14
Groups 2 and 13
Groups 2 and 14
Advertisements
उत्तर
Groups 2, 13 and 14
Explanation:
Cement contains elements of group 2 (Ca), group 13 (Al) and group 14 (Si).
APPEARS IN
संबंधित प्रश्न
How can you explain higher stability of BCl3 as compared to TlCl3?
If B–Cl bond has a dipole moment, explain why BCl3 molecule has zero dipole moment.
Aluminium trifluoride is insoluble in anhydrous HF but dissolves on the addition of NaF. Aluminium trifluoride precipitates out of the resulting solution when gaseous BF3 is bubbled through. Give reasons.
How would you explain the lower atomic radius of Ga as compared to Al?
In some of the reactions thallium resembles aluminium, whereas in others it resembles with group I metals. Support this statement by giving some evidences.
Which of the following oxides is acidic in nature?
The exhibition of highest co-ordination number depends on the availability of vacant orbitals in the central atom. Which of the following elements is not likely to act as central atom in \[\ce{MF^{3-}6}\]?
In the structure of diborane ______.
Which of the following statements are correct. Answer on the basis of Figure.
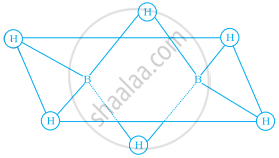
(i) The two birdged hydrogen atoms and the two boron atoms lie in one plane;
(ii) Out of six B – H bonds two bonds can be described in terms of 3 centre 2-electron bonds.
(iii) Out of six B – H bonds four B – H bonds can be described in terms of 3 centre 2 electron bonds;
(iv) The four-terminal B – H bonds are two centre-two electron regular bonds.
Explain why the following compounds behave as Lewis acids?
BCl3
Explain why the following compounds behave as Lewis acids?
AlCl3
Explain the following:
Pb4+ acts as an oxidising agent but Sn2+ acts as a reducing agent.
Describe the general trends in the following properties of the elements in Groups 13 and 14.
Metallic character
Describe the general trends in the following properties of the elements in Groups 13 and 14.
Oxidation states
Account for the following observations:
The +1 oxidation state of thallium is more stable than its +3 state.
Three pairs of compounds are given below. Identify that compound in each of the pairs which has group 13 element in more stable oxidation state. Give reason for your choice. State the nature of bonding also.
AlCl3 , AlCl
Three pairs of compounds are given below. Identify that compound in each of the pairs which has group 13 element in more stable oxidation state. Give reason for your choice. State the nature of bonding also.
InCl3, InCl
Boron fluoride exists as BF3 but boron hydride doesn’t exist as BH3. Give reason. In which form does it exist? Explain its structure.
