Advertisements
Advertisements
प्रश्न
At what temperature is the root mean square speed of an atom in an argon gas cylinder equal to the rms speed of a helium gas atom at – 20 °C? (atomic mass of Ar = 39.9 u, of He = 4.0 u).
Advertisements
उत्तर १
Temperature of the helium atom, THe = –20°C= 253 K
Atomic mass of argon, MAr = 39.9 u
Atomic mass of helium, MHe = 4.0 u
Let, (vrms)Ar be the rms speed of argon.
Let (vrms)He be the rms speed of helium.
The rms speed of argon is given by:
`(v_"rms")_"Ar" = sqrt((3RT_"Ar")/M_"Ar")` .... (1)
Where,
R is the universal gas constant
TAr is temperature of argon gas
The rms speed of helium is given by:
`(v_"rms")_"He" = sqrt((3RT_"He")/M_"He")` ... (ii)
It is given that
(vrms)Ar = (vrms)He
`sqrt((3RT_"Ar")/ M_"Ar")` = `sqrt((3RT_"He")/M_"He")`
`T_"Ar"/M_"Ar" = T_"He"/M_"He"`
`T_"Ar" = T_"He"/M_"He" xx M_"Ar"`
`= 253/4 xx 39.9`
= 2523.675 = 2.52 × 103 K
Therefore, the temperature of the argon atom is 2.52 × 103 K.
उत्तर २
Let C and C’ be the rms velocity of argon and a helium gas atoms at temperature T K and T K respectively
Here, M = 39.9; M’ = 4.0; T =?; T = -20 + 273 = 253 K
Now, `C = sqrt((3RT)/M
)= sqrt((3RT)/39.9)` and `C' = sqrt((3RT')/M') = sqrt(3R xx 253)/4`
Since C =C'
Therefore `sqrt((3RT)/39.9) = sqrt(3Rxx253)/4` or `T = (39.9 xx 253)/4 = 2523.7 K`
संबंधित प्रश्न
The figure shows the plot of PV/T versus Pfor 1.00×10–3 kg of oxygen gas at two different temperatures.

(a) What does the dotted plot signify?
(b) Which is true: T1 > T2 or T1 < T2?
(c) What is the value of PV/T where the curves meet on the y-axis?
(d) If we obtained similar plots for 1.00 ×10–3 kg of hydrogen, would we get the same value of PV/T at the point where the curves meet on the y-axis? If not, what mass of hydrogen yields the same value of PV/T (for low pressure high temperature region of the plot)? (Molecular mass of H2 = 2.02 u, of O2 = 32.0 u, R = 8.31 J mo1–1 K–1.)
Three vessels of equal capacity have gases at the same temperature and pressure. The first vessel contains neon (monatomic), the second contains chlorine (diatomic), and the third contains uranium hexafluoride (polyatomic).
Is the root mean square speed of molecules the same in the three cases? If not, in which case is vrms the largest?
50 m3 of saturated vapour is cooled down from 30°C to 20°C. Find the mass of the water condensed. The absolute humidity of saturated water vapour is 30 g m−3 at 30°C and 16 g m−3 at 20°C.
What do you understand by gas?
What is diffusion? Give an example to illustrate it.
Give reasons for the following:
Gas fills the vessel completely in which it is kept.
Match the following:
|
|
Column A |
Column B |
|
(a) |
cm3 |
(i) Pressure |
|
(b) |
Kelvin |
(ii) Temperature |
|
(c) |
Torr |
(iii) Volume |
|
(d) |
Boyle's law |
(iv) `"V"/"T" = ("V"_1)/("T"_1)` |
|
(a) |
Charles's law |
(v) `"PV"/"T" = ("P"_1 "V"_1)/"T"_1` |
|
|
|
(vi) PV = P1V1 |
Name or state the following:
An equation used in chemical calculations which gives a simultaneous effect of changes of temperature and pressure on the volume of a given mass of dry gas
Give reason for the following:
Temperature remaining constant the product of the vol. & the press, of a given mass of dry gas is a constant.
Show that for monoatomic gas the ratio of the two specific heats is 5:3.
Show that for diatomic gas the ratio of the two specific heats is 7:5.
Gases exert pressure on the walls of the container because the gas molecules ______
The equation of state for 2g of oxygen at a pressure 'P' and temperature 'T', when occupying a volume 'V' will be ______.
Estimate the average thermal energy of a helium atom at the temperature on the surface of the Sun (6000 K).
Three vessels of equal capacity have gases at the same temperature and pressure. The first vessel contains neon (monatomic), the second contains chlorine (diatomic), and the third contains uranium hexafluoride (polyatomic).
Do the vessels contain an equal number of respective molecules?
Which of the following diagrams (Figure) depicts ideal gas behaviour?
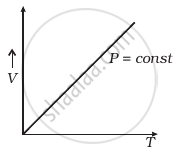 (a) |
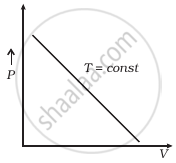 (b) |
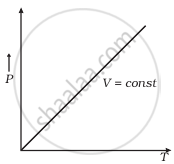 (c) |
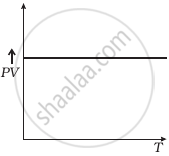 (d) |
Calculate the number of atoms in 39.4 g gold. Molar mass of gold is 197g mole–1.
