Advertisements
Advertisements
प्रश्न
Match the following:
|
|
Column A |
Column B |
|
(a) |
cm3 |
(i) Pressure |
|
(b) |
Kelvin |
(ii) Temperature |
|
(c) |
Torr |
(iii) Volume |
|
(d) |
Boyle's law |
(iv) `"V"/"T" = ("V"_1)/("T"_1)` |
|
(a) |
Charles's law |
(v) `"PV"/"T" = ("P"_1 "V"_1)/"T"_1` |
|
|
|
(vi) PV = P1V1 |
Advertisements
उत्तर
|
Column A |
Column B |
|
(a) cm3 |
Volume |
|
(b) Kelvin |
Temperature |
|
(c) Torr |
Pressure |
|
(d) Boyle's law |
PV = P1 V1
|
|
(e) Charles's law |
`"V"/"T" = ("V"_1)/("T"_1)` |
APPEARS IN
संबंधित प्रश्न
Molar volume is the volume occupied by 1 mol of any (ideal) gas at standard temperature and pressure (STP: 1 atmospheric pressure, 0 °C). Show that it is 22.4 litres
50 m3 of saturated vapour is cooled down from 30°C to 20°C. Find the mass of the water condensed. The absolute humidity of saturated water vapour is 30 g m−3 at 30°C and 16 g m−3 at 20°C.
Give reasons for the following:
Gas fills the vessel completely in which it is kept.
A gas occupies 500 cm3 at a normal temperature. At what temperature will the volume of the gas be reduced by 20% of its original volume, the pressure is constant?
Gases exert pressure on the walls of the container because the gas molecules ______
Which of the following diagrams (Figure) depicts ideal gas behaviour?
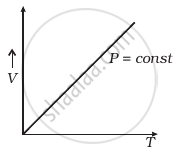 (a) |
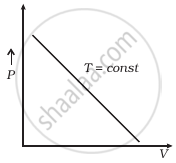 (b) |
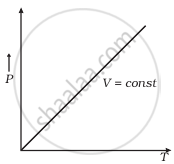 (c) |
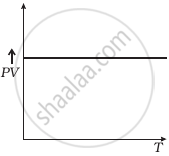 (d) |
Calculate the number of atoms in 39.4 g gold. Molar mass of gold is 197g mole–1.
At room temperature, a diatomic gas is found to have an r.m.s. speed of 1930 ms-1. The gas is ______.
For a wave, y = 0.0002 sin`[2pi(110"t"-x/3)+pi/3]` is travelling in a medium. The energy per unit volume being transferred by wave if density of medium is 1.5 kg/m3, is ______.
P ∝ T at constant volume is the statement of ______.
