Advertisements
Advertisements
प्रश्न
Calculate the number of atoms in 39.4 g gold. Molar mass of gold is 197g mole–1.
Advertisements
उत्तर
We know that,
Molar mass = Mass of Avogadro's number of atoms (Molecules).
= 6.023 × 1023 atoms.
Given, molar mass of gold = 197 g/mol
Now,
∴ 197 g of gold contains = 6.023 × 1023 atoms
∴ 1 g of gold contain = `(6.023 xx 10^23)/197` atoms
∴ 39.4 g of gold contains = `(6.023 xx 10^23 xx 39.4)/197`
⇒ 1.20 × 1023 atoms.
APPEARS IN
संबंधित प्रश्न
Give reasons for the following:
Gas fills the vessel completely in which it is kept.
Choose the correct answer:
The graph of PV vs P for gas is
Match the following:
|
|
Column A |
Column B |
|
(a) |
cm3 |
(i) Pressure |
|
(b) |
Kelvin |
(ii) Temperature |
|
(c) |
Torr |
(iii) Volume |
|
(d) |
Boyle's law |
(iv) `"V"/"T" = ("V"_1)/("T"_1)` |
|
(a) |
Charles's law |
(v) `"PV"/"T" = ("P"_1 "V"_1)/"T"_1` |
|
|
|
(vi) PV = P1V1 |
Fill in the blanks:
If the temperature is reduced to half, ………….. would also reduce to half.
A gas occupies 500 cm3 at a normal temperature. At what temperature will the volume of the gas be reduced by 20% of its original volume, the pressure is constant?
Correct the following statement:
0°C is equal to zero Kelvin.
Name or state the following:
The absolute temperature value corresponding to 35°C.
Show that for diatomic gas the ratio of the two specific heats is 7:5.
Estimate the average thermal energy of a helium atom at room temperature (27 °C).
Estimate the average thermal energy of a helium atom at the temperature on the surface of the Sun (6000 K).
Three vessels of equal capacity have gases at the same temperature and pressure. The first vessel contains neon (monatomic), the second contains chlorine (diatomic), and the third contains uranium hexafluoride (polyatomic).
Do the vessels contain an equal number of respective molecules?
Which of the following diagrams (Figure) depicts ideal gas behaviour?
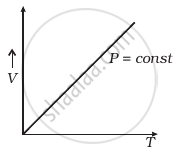 (a) |
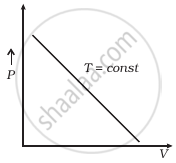 (b) |
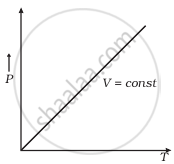 (c) |
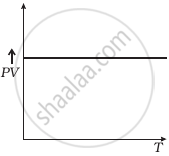 (d) |
A box of 1.00 m3 is filled with nitrogen at 1.50 atm at 300K. The box has a hole of an area 0.010 mm2. How much time is required for the pressure to reduce by 0.10 atm, if the pressure outside is 1 atm.
Cooking gas containers are kept in a lorry moving with uniform speed. The temperature of the gas molecules inside will ______.
At room temperature, a diatomic gas is found to have an r.m.s. speed of 1930 ms-1. The gas is ______.
