Advertisements
Advertisements
प्रश्न
Show that for monoatomic gas the ratio of the two specific heats is 5:3.
Advertisements
उत्तर
- For a monatomic gas enclosed in a container, held at a constant temperature T, and containing NA atoms, each atom has only 3 translational degrees of freedom.
- Therefore, the average energy per atom is `3/2` kBT and the total internal energy per mole is, E = `3/2` NAkBT
- Molar specific heat at constant volume
Cv = `"dE"/"dT" = 3/2"N"_"A""k"_"B" = 3/2"R"` - Using Mayer’s relation, CP = R + CV
∴ Cp = `5/2`R
∴ `"C"_"p"/"C"_"v" = 5/3`
APPEARS IN
संबंधित प्रश्न
Molar volume is the volume occupied by 1 mol of any (ideal) gas at standard temperature and pressure (STP: 1 atmospheric pressure, 0 °C). Show that it is 22.4 litres
The figure shows the plot of PV/T versus Pfor 1.00×10–3 kg of oxygen gas at two different temperatures.

(a) What does the dotted plot signify?
(b) Which is true: T1 > T2 or T1 < T2?
(c) What is the value of PV/T where the curves meet on the y-axis?
(d) If we obtained similar plots for 1.00 ×10–3 kg of hydrogen, would we get the same value of PV/T at the point where the curves meet on the y-axis? If not, what mass of hydrogen yields the same value of PV/T (for low pressure high temperature region of the plot)? (Molecular mass of H2 = 2.02 u, of O2 = 32.0 u, R = 8.31 J mo1–1 K–1.)
At what temperature is the root mean square speed of an atom in an argon gas cylinder equal to the rms speed of a helium gas atom at – 20 °C? (atomic mass of Ar = 39.9 u, of He = 4.0 u).
50 m3 of saturated vapour is cooled down from 30°C to 20°C. Find the mass of the water condensed. The absolute humidity of saturated water vapour is 30 g m−3 at 30°C and 16 g m−3 at 20°C.
What do you understand by gas?
Give reasons for the following:
Gas fills the vessel completely in which it is kept.
Choose the correct answer:
The graph of PV vs P for gas is
Match the following:
|
|
Column A |
Column B |
|
(a) |
cm3 |
(i) Pressure |
|
(b) |
Kelvin |
(ii) Temperature |
|
(c) |
Torr |
(iii) Volume |
|
(d) |
Boyle's law |
(iv) `"V"/"T" = ("V"_1)/("T"_1)` |
|
(a) |
Charles's law |
(v) `"PV"/"T" = ("P"_1 "V"_1)/"T"_1` |
|
|
|
(vi) PV = P1V1 |
Fill in the blanks:
If the temperature is reduced to half, ………….. would also reduce to half.
A gas occupies 500 cm3 at a normal temperature. At what temperature will the volume of the gas be reduced by 20% of its original volume, the pressure is constant?
Name or state the following:
The absolute temperature value corresponding to 35°C.
The average energy per molecule is proportional to ______
If the absolute temperature of a body is doubled, the power radiated will increase by a factor of ______
Gases exert pressure on the walls of the container because the gas molecules ______
Estimate the average thermal energy of a helium atom at room temperature (27 °C).
Estimate the average thermal energy of a helium atom at the temperature on the surface of the Sun (6000 K).
Three vessels of equal capacity have gases at the same temperature and pressure. The first vessel contains neon (monatomic), the second contains chlorine (diatomic), and the third contains uranium hexafluoride (polyatomic).
Do the vessels contain an equal number of respective molecules?
Which of the following diagrams (Figure) depicts ideal gas behaviour?
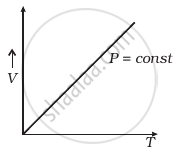 (a) |
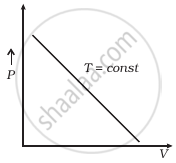 (b) |
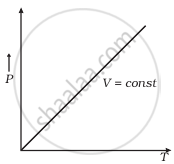 (c) |
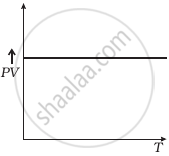 (d) |
Calculate the number of atoms in 39.4 g gold. Molar mass of gold is 197g mole–1.
A box of 1.00 m3 is filled with nitrogen at 1.50 atm at 300K. The box has a hole of an area 0.010 mm2. How much time is required for the pressure to reduce by 0.10 atm, if the pressure outside is 1 atm.
The volume V of an enclosure contains a mixture of three gases, 16 g of oxygen, 28 g of nitrogen and 44 g of carbon dioxide at absolute temperature T. Consider R as universal gas constant. The pressure of the mixture of gases is ______.
For a wave, y = 0.0002 sin`[2pi(110"t"-x/3)+pi/3]` is travelling in a medium. The energy per unit volume being transferred by wave if density of medium is 1.5 kg/m3, is ______.
P ∝ T at constant volume is the statement of ______.
