Advertisements
Advertisements
प्रश्न
Arrange the following compounds in increasing order of their property as indicated:
CH3CH2CH(Br)COOH, CH3CH(Br)CH2COOH, (CH3)2CHCOOH, CH3CH2CH2COOH (acid strength)
Advertisements
उत्तर
Carboxylic acid molecules with electron-withdrawing groups or electronegative atoms are more acidic. In addition, bonding the atom/group further away from the a-carbon reduces acidity. Thus, the acid strength is:
\[\begin{array}{cc}
\phantom{.....................................}\ce{Br}\phantom{.......................}\ce{Br}\\
\phantom{....................................}|\phantom{.........................}|\\
\ce{\underset{I}{(CH3)2CHCOOH} < \underset{II}{CH3(CH2)2COOH} < \underset{III}{CH3CHCH2COOH} < \underset{IV}{CH3CH2CHCOOH}}\
\end{array}\]
The lesser number of −CH3 groups on the α-carbon in II results in increased acidity compared to I. The −CH3 group increases electron density through the +I action on α-carbon which also affects the −COOH group, hindering the release of H+.
APPEARS IN
संबंधित प्रश्न
How is 1-nitropropane prepared from suitable oxime?
How are 2 - nitropropane prepared from suitable oxime?
Distinguish between the following : Benzoic acid and methyl benzoate
CH3CO2H or CH2FCO2H
Which acid of the pair shown here would you expect to be stronger?
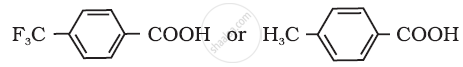
Write the reactions involved Hell-Volhard Zelinsky reaction?
Account for the following:
Carboxylic acid is a stronger acid than phenol.
What happens when Salicylic acid is treated with (CH3CO)2 O/H+?
Arrange the following compounds in increasing order of their property as indicated:
F - CH2COOH, O2N - CH2 COOH CH3 COOH,HCOOH - acid character.
Assertion: Formaldehyde is a planar molecule.
Reason: It contains sp2 hybridised carbon atom.
Acidity of BF3 can be explained on the basis of which of the following concepts?
A mixture of benzaldehyde and formaldehyde on heating with 50% NaOH solution gives
When propionamide reacts with Br2 in the presence of alkali the product is ______.
Formic acid and formaldehyde can be distinguished by treating with ______.
Describe the action of alcoholic potassium hydroxide (alc. KOH) on ethyl bromide
Describe the action of alcoholic potassium hydroxide (alc. KOH) on n-propyl bromide
Describe the action of alcoholic potassium hydroxide (alc. KOH) on isopropyl bromide.
