Advertisements
Advertisements
प्रश्न
Arrange the following compounds in increasing order of their property as indicated:
F - CH2COOH, O2N - CH2 COOH CH3 COOH,HCOOH - acid character.
Advertisements
उत्तर
F - CH2COOH, O2N - CH2 COOH CH3 COOH,HCOOH - acid character.
CH3COOH < HCOOH < F - CH2 COOH < NO2 CH COOH
Methyl group in CH3 COOH decreases acidity due to its electron withdrawing nature, hence less acidic than formic acid whereas electron withdrawing group, group NO2 and F shows greater acidity with FCH2 COOH highest due to strongly electronegative fluorine group
APPEARS IN
संबंधित प्रश्न
How are 2 - nitropropane prepared from suitable oxime?
Although phenoxide ion has more number of resonating structures than carboxylate ion, carboxylic acid is a stronger acid than phenol. Give two reasons.
Why pKa of F-CH2-COOH is lower than that of Cl−CH2−COOH?
Which acid of the pair shown here would you expect to be stronger?
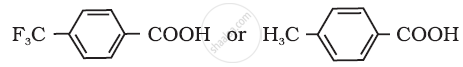
Arrange the following compounds in increasing order of their property as indicated:
CH3CH2CH(Br)COOH, CH3CH(Br)CH2COOH, (CH3)2CHCOOH, CH3CH2CH2COOH (acid strength)
Complete the following reaction sequence.
\[\begin{array}{cc}
\ce{O}\phantom{...............................................}\\
||\phantom{...............................................}\\
\ce{CH3 - C - CH3 ->[(i) CH3MgBr][H2O] (A) ->[Na metal][Ether] (B) ->[CH3 - Br] (C)}
\end{array}\]
Acidity of BF3 can be explained on the basis of which of the following concepts?
When propionamide reacts with Br2 in the presence of alkali the product is ______.
Describe the action of alcoholic potassium hydroxide (alc. KOH) on ethyl bromide
The correct order of decreasing acidity of the following aliphatic acids is:
