Advertisements
Advertisements
प्रश्न
Arrange the following compounds in increasing order of reactivity towards SN2 reaction.
2-Bromopentane, 1-Bromopentane, 2-Bromo-2-methylbutane
Advertisements
उत्तर
SN2 reaction involves the formation of transition state. Therefore, reactivity towards SN2 depends upon the steric hindrance:
Due to the presence of 2 methyl group in 2-Bromo-2-methylbutane it is least reactive towards SN2 reactions, 2-bromopentane is moderately reactive while 1-bromopentane is most reactive.
2-Bromo-2-methylbutane < 2-Bromopentane < 1-Bromopentane
APPEARS IN
संबंधित प्रश्न
Out of  , which is more reactive towards SN1 reaction and why?
, which is more reactive towards SN1 reaction and why?
What is the action of the following on ethyl bromide
alcoholic solution of potassium hydroxide.
In a coordination entity of the type [PtCl2(en)2]2+ which isomer will show optical isomerism?
SN1 reaction of alkyl halides lead to ___________.
Which of the following compound will undergo racemisation when reacts with aq. KOH?
(i)
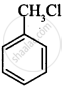
(ii)
CH3CH2CH2Cl
(iii)
\[\begin{array}{cc}
\ce{CH3}\phantom{..}\\
|\phantom{....}\\
\ce{CH3-CH-CH2Cl}
\end{array}\]
(iv)
\[\begin{array}{cc}
\phantom{..}\ce{H}\\
\phantom{..}|\\
\ce{CH3-C-Cl}\\
\phantom{..}|\\
\phantom{.....}\ce{C2H5}
\end{array}\]
The increasing order of reactivity towards SN1 mechanism is:
(I) \[\begin{array}{cc}
\ce{CH3-CH-CH2-CH3}\\
|\phantom{........}\\
\ce{CH3}\phantom{.....}
\end{array}\]
(II) CH3CH2CH2Cl
(III) P–CH3O–C6H4–CH2Cl
Which of the following alkyl halides will undergo SN1 reaction most readily?
Ethylene chloride and ethylidene chloride are isomers. Identify the correct statements.
(i) Both the compounds form same product on treatment with alcoholic KOH.
(ii) Both the compounds form same product on treatment with aq.NaOH.
(iii) Both the compounds form same product on reduction.
(iv) Both the compounds are optically active.
In which reaction mechanism carbocation is formed?
The number of chiral alcohol (s) with molecular formula C4H10O is ______.
