Topics
Solid State
- Introduction to Solid State
- Amorphous and Crystalline Solids
- Crystal Lattices and Unit Cells
- Calculations Involving Unit Cell Dimensions
- Close Packed Structures of Solids
- Efficiency of Packing in Body-centred Cubic Structures
- Close Packed Structures - Formula of a Compound and Number of Voids Filled
- Number of Atoms in a Unit Cell
- Imperfections in Solids - Introduction
- Properties of Solids: Magnetic Properties
- Band Theory of Metals
- Properties of Solids: Electrical Properties
- Applications of n-type and p-type Semiconductors
Solid State
Solutions and Colligative Properties
- Types of Solutions
- Expressing Concentration of Solutions
- Solubility of a Gas in a Liquid
- Solubility of a Solid in a Liquid
- Colligative Properties and Determination of Molar Mass
- Relative Lowering of Vapour Pressure
- Depression of Freezing Point
- Osmosis and Osmotic Pressure
- Abnormal Molar Masses
- Elevation of Boiling Point
Solutions
- Introduction to Solutions
- Types of Solutions
- Capacity of Solution to Dissolve Solute
- Solubility
- Vapour Pressure of Solutions of Liquids in Liquids
- Colligative Properties of Nonelectrolyte Solutions
- Vapour Pressure Lowering
- Boiling Point Elevation
- Depression in Freezing Point
- Osmosis and Osmotic Pressure
- Colligative Properties of Electrolytes
- Overview of Solutions
Ionic Equilibria
Chemical Thermodynamics and Energetic
- Chemical Thermodynamics and Energetic
- Concepts of System
- Types of Systems
- Surroundings
- Work, Heat, Energy, Extensive and Intensive Properties
- State Functions
- First Law of Thermodynamics
- Internal Energy and Enthalpy
- Hess’ Law of Constant Heat Summation
- Enthalpy of Bond Dissociation
- Combustion, Formation, Atomization, Sublimation
- Phase Transition
- Ionization and Solution
- Dilution Introduction of Entropy as a State Function
- Free Energy Change for Spontaneous and Non Spontaneous Processes
- Equilibrium Constant
- Second and Third Law of Thermodynamics
Electrochemistry
- Electrochemical Cells
- Conductance of Electrolytic Solutions - Introduction
- Variation of Conductivity and Molar Conductivity with Concentration
- Electrolytic Cells and Electrolysis - Introduction
- Primary Batteries
- Lead Accumulator
- Galvanic or Voltaic Cell
- Nernst Equation
- Relation Between Gibbs Energy Change and Emf of a Cell
- fuel cell
- Factors Affecting Corrosion
- Concept of Redox Reactions
Chemical Thermodynamics
- Chemical Thermodynamics
- Terms Used in Thermodynamics
- Nature of Heat and Work
- Expression for Pressure-volume (PV) Work
- Concept of Maximum Work
- Internal Energy (U)
- First Law of Thermodynamics
- Enthalpy (H)
- Enthalpies of Physical Transformations
- Thermochemistry
- Spontaneous (Irreversible) Process
- Overview of Chemical Thermodynamics
Electrochemistry
- Introduction to Electrochemistry
- Electric Conduction
- Electrical Conductance of Solution
- Electrochemical Cells
- Electrolytic Cells
- Galvanic or Voltaic Cell
- Electrode Potential and Cell Potential
- Thermodynamics of Galvanic Cells
- Reference Electrodes
- Galvanic Cells Useful in Day-to-day Life
- Fuel Cells
- Electrochemical Series (Electromotive Series)
- Overview of Electrochemistry
Chemical Kinetics
Chemical Kinetics
- Concept of Chemical Kinetics
- Rate of Reactions
- Rate of Reaction and Reactant Concentration
- Molecularity of Reaction
- Integrated Rate Equations
- First Order Reactions
- Zero Order Reactions
- Half Life Period of a Reaction
- Pseudo First Order Reaction
- Integrated Rate Law for Gas Phase f Reactions
- Collision Theory of Bimolecular Reactions
- Temperature Dependence of Reaction Rates
- Effect of a Catalyst on the Rate of Reaction
- Overview of Chemical Kinetics
General Principles and Processes of Isolation of Elements
P-block Elements
Elements of Groups 16, 17 and 18
- Occurrence of Elements of Groups 16, 17 and 18
- Electronic Configuration of Elements of Group 16, 17 and 18
- Atomic and Physical Properties of Elements of Group 16, 17 and 18
- Anomalous Behaviour of Oxygen
- Anomalous Behaviour of Fluorine
- Chemical Properties of Elements of Groups 16, 17 and 18
- Oxoacids
- Oxygen and Compounds of Oxygen
- Chlorine
- Group 18 Elements - The Noble gas Family
- Overview of Elements of Groups 16, 17 and 18
Group 15 Elements
Group 16 Elements
Group 17 Elements
Group 18 Elements
Transition and Inner Transition Elements
- Transition and Inner Transition Elements
- Position in the Periodic Table of Transition and Inner Transition Elements
- Electronic Configuration of Transition and Inner Transition Elements
- Oxidation States of First Transition Series
- Physical Properties of First Transition Series
- Trends in Atomic Properties of the First Transition Series
- Preparation of Potassium Permanganate
- Chemical Properties of KMnO4
- Uses of KMnO4
- K2Cr2O7: Preparation of Potassium Dichromate
- Chemical Properties of K2Cr2O7
- Common Properties of d Block Elements
- Basic Principles of Metallurgy > Extraction of Metals
- Inner Transition (f-block) Elements: Lanthanoids and Actinoids
- Properties of f-block Elements
- Properties of Lanthanoids
- Applications of Lanthanoids
- The Actinoids
- Properties of Actinoids
- Applications of Actinoids
- Postactinoid Elements
- Overview of Transition and Inner Transition Elements
D and F Block Elements
D-block Elements
- General Indroduction
- Position in the Periodic Table - d-block Elements
- Electronic Configurations of the d-block Elements
- General Properties of the Transition Elements (d-block)
- Some Important Compounds of Transition Elements
F-block Elements
Coordination Compounds
- Concept of Coordination Compounds
- Types of Ligands
- Terms Used in Coordination Chemistry
- Classification of Complexes
- IUPAC Nomenclature of Coordination Compounds
- Effective Atomic Number (EAN) Rule
- Isomerism in Coordination Compounds
- Stability of the Coordination Compounds
- Theories of Bonding in Complexes
- Applications of Coordination Compounds
- Overview of Coordination Compounds
Coordination Compounds
- Introduction
- Nomenclature of Coordination Compounds
- Importance of Coordination Compounds
- Important Terms Pertaining to Coordination Compounds
- Colour in Coordination Compounds
- Magnetic Properties of Coordination Compounds
- Bonding in Coordination Compounds
- Werner’s Theory of Coordination Compounds
- Valence Bond Theory (VBT)
- Crystal Field Theory (CFT)
- Stereoisomerism
- Structural Isomerism
- Importance of Coordination Compounds
Halogen Derivatives of Alkanes (And Arenes)
Halogen Derivatives
- Classification of Halogen Derivatives
- Nomenclature of Halogen Derivatives
- Methods of Preparation of Alkyl Halides
- Physical Properties of Halogen Derivatives
- Optical Isomerism in Halogen Derivatives
- Chemical Properties of Halogen Derivatives
- Reaction with Active Metals
- Polyhalogen Compounds
- Nomenclature
- Reactions of Haloalkanes - Elimination Reactions
- Overview of Halogen Derivatives
Haloalkanes
- Introduction to Haloalkanes and Haloarenes
- Nomenclature
- Nature of C-X Bond
- Physical Properties of Haloalkanes and Haloarenes
- Methods of Preparation of Haloalkanes and Haloarenes
- Reactions of Haloalkanes - Nucleophilic Substitution Reactions
- Reactions of Haloalkanes - Elimination Reactions
- R-s and D-l Configuration
Haloarenes
- Introduction to Haloalkanes and Haloarenes
- Nature of C-X Bond
- Reactions of Haloarenes - Nucleophilic Substitution
- Nucleophilic Substitution
- Electrophilic Substitution Reactions
- Reaction with Metals
- R-S and D-l Configurations
- Polyhalogen Compounds
Alcohols, Phenols and Ethers Alcohols
Alcohols, Phenols and Ethers
Alcohols
Phenols
- Phenols
- Nomenclature
- Methods of Preparation
- Physical and Chemical Properties
- Acidic Nature of Phenol
- Electrophillic Substitution Reactions
- Uses of Phenols
Ethers
Aldehydes, Ketones and Carboxylic Acids
Aldehydes, Ketones and Carboxylic Acids
- Concepts of Aldehydes, Ketones, and Carboxylic Acids
- Classification of Aldehydes, Ketones and Carboxylic Acids
- Nomenclature of Aldehydes, Ketones and Carboxylic Acids
- Preparation of Aldehydes and Ketones
- Preparation of Carboxylic Acids
- Physical Properties
- Polarity of Carbonyl Group
- Chemical Properties of Aldehydes and Ketones
- Chemical Properties of Carboxylic Acids
- Chemical Reactions of Aldehydes and Ketones - Reactions Due to α-hydrogen
- Overview: Aldehydes, Ketones and Carboxylic Acids
- Overview of Aldehydes, Ketones and Carboxylic Acids
Aldehydes and Ketones
- Concepts of Aldehydes, Ketones, and Carboxylic Acids
- Nomenclature of Aldehydes and Ketones
- Nature of Carbonyl Group
- Preparation of Aldehydes and Ketones
- Physical Properties of Aldehydes and Ketones
- Chemical Reactions of Aldehydes and Ketones - Nucleophilic Addition Reactions
- Reactivity of Alpha Hydrogen in Aldehydes
- Uses of Aldehydes and Ketones
- Chemical Reactions of Aldehydes and Ketones - Other Reactions
Carboxylic Acids
- Carboxylic Acids
- Nomenclature of Carboxylic Acids
- Structure of the Carboxyl group
- Methods of Preparation of Carboxylic Acids
- Physical Properties of Carboxylic Acids
- Chemical Properties and Reactions of Carboxylic Acid
- Chemical Reactions of Carboxylic Acids - Reactions Involving Cleavege of O-H Bond
- Chemical Reactions of Carboxylic Acids - Reactions Involving Cleavege of C-OH Bond
- Chemical Reactions of Carboxylic Acids - Reactions Involving –COOH Group
- Chemical Reactions of Carboxylic Acids - Substitution Reactions in the Hydrocarbon Part
- Uses of Carboxylic Acids
Organic Compounds Containing Nitrogen
Amines
Amines
- Concept of Amines
- Nomenclature of Animes
- Classification of Amines
- Structure of Amines
- Physical Properties of Amines
- Uses of Amines
- Identification of Primary, Secondary and Tertiary Amines
- Secondary and Tertiary Amines
- Chemical Reactions of Amines - Carbylamine Reaction
- Chemical Reactions of Amines - Reaction with Nitrous Acid
- Chemical Reactions of Amines - Reaction with Arylsulphonyl Chloride
- Chemical Reactions of Amines - Electrophilic Substitution
- Preparation of Amines
Cyanides and Isocyanides
Diazonium Salts
Biomolecules
- Principal Molecules of the Living World
- Biomolecules in the Cell > Carbohydrates
- Biomolecules in the Cell > Proteins
- Overview of Biomolecules
- Overview: Biomolecules
Biomolecules
Carbohydrates
Proteins
- Introduction of Proteins
- Amino Acids
- Peptide
- Linkage
- Polypeptides
- Structure of Proteins
- Biomolecules in the Cell > Enzymes
- Biomolecules in the Cell > Hormones
- Structure, Classification and Functions
Vitamins
- Biomolecules in the Cell > Proteins
- Classification of Vitamins
- Important Vitamins, Their Sources and Their Deficiency Diseases
Nucleic Acids
Introduction to Polymer Chemistry
- Polymer Chemistry
- Classification of Polymers> Based On Type of Different Monomers
- Some Important Polymers
- Molecular Mass and Degree of Polymerization of Polymers
- Classification of Polymers> Based on Biodegradability
- Commercially Important Polymers
- Preparation of Polytetrafluoroethene (Teflon)
- Overview of Introduction to Polymer Chemistry
Polymers
- Introduction to Polymers
- Classification of Polymers> Based on Source or Origin
- Types of Polymerisation Reactions - Addition Polymerisation or Chain Growth Polymerisation
- Types of Polymerisation Reactions - Copolymerisation
- Some Important Polymers
- Types of Polymerisation Reactions - Condensation Polymerisation Or Step Growth Polymerisation
- Nylon 6
- Nylon 66
- Types of Polymerisation Reactions - Rubber
- Classification of Polymers> Based on Biodegradability
- Preparation of Polytetrafluoroethene (Teflon)
- Preparation of Polyacrylonitrile
Chemistry in Everyday Life
Green Chemistry and Nanochemistry
- Green Chemistry and Nanochemistry
- Principles of Green Chemistry
- The Role of Green Chemistry
- Introduction to Nanochemistry
- Characteristic Features of Nanoparticles
- Synthesis of Nanomaterials
- History of Nanotechnology
- Applications of Nanomaterials
- Nanoparticles and Nanotechnology
- Overview of Green Chemistry and Nanochemistry
Chemicals in Medicines
Chemicals in Food
Cleansing Agents
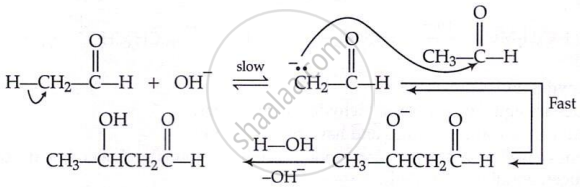
Key Points: Chemical Reactions of Aldehydes and Ketones - Reactions Due to α-hydrogen
Aldol condensation:
\[\ce{CH3 - \overset{\displaystyle O}{\overset{||}{C}} - H + H - CH2 - \overset{\displaystyle O}{\overset{||}{C}} - H} \xrightarrow{\text{dil. NaOH}} \ce{H3C - \underset{\underset{\displaystyle OH}{|}}{CH} - \underset{\underset{\displaystyle H}{|}}{CH} - \overset{\displaystyle O}{\overset{||}{C}} - H} \\ \xrightarrow{\text{dil. }\ce{H2SO4}\Delta, \ce{-H2O}} \ce{CH3 - CH = CH - \overset{\displaystyle O}{\overset{||}{C}} - H}\]
Mechanism:
Cross aldol condensation:
\[\begin{array}{ccccccccccccc} & \ce{O} & & \ce{O} & & & & \ce{OH} & & \ce{O} & & & \\ & || & & || & & & & | & & || & & & \\ \ce{C6H5 -} & \ce{C} & \ce{+ HCH2 -} & \ce{C} & \ce{- H} & \overset{\text{dil. NaOH}}{\rightleftharpoons} & \ce{C6H5 -} & \ce{C} & \ce{- CH2 -} & \ce{C} & \ce{- H} & \xrightarrow[\Delta]{\ce{H2O}} & \ce{C6H5CH=CHCHO} \\ & | & & & & & & | & & & & & \text{Cinnamaldehyde} \\ & \ce{H} & & & & & & \ce{H} & & & & & \end{array}\]
Cannizzaro reaction: It is a self-oxidation reduction reaction.
\[\begin{array}{r@{\;}c@{\;}l} \ce{H} & & \\ & \backslash & \\ & & \ce{C=O} \\ & / & \\ \ce{H} & & \end{array} + \begin{array}{r@{\;}c@{\;}l} \ce{H} & & \\ & \backslash & \\ & & \ce{C=O} \\ & / & \\ \ce{H} & & \end{array} + \text{conc. KOH} \xrightarrow{\Delta} \ce{H - \underset{\underset{\displaystyle H}{|}}{\overset{\overset{\displaystyle H}{|}}{C}} - OH} + \ce{H - \overset{\displaystyle O}{\overset{||}{C}} - OK}\]
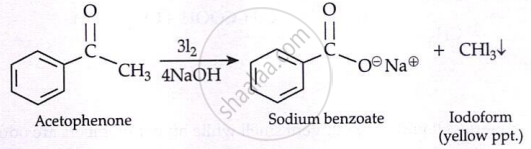
Haloform Reaction:
The reaction can be used to transform acetyl groups into carboxyl groups or to produce chloroform or iodoform. This reaction has been used in qualitative analysis to indicate the presence of a methyl ketone in which excess iodine is used to halogenate the compound. The product iodoform precipitates as a yellow-coloured substance and has a characteristic odour.
\[\underset{\substack{\text{Acetone} \\ \text{Iodoform Test}}}{\ce{H3C - \overset{\displaystyle O}{\overset{||}{C}} - CH3}} \xrightarrow[4\ce{NaOH}]{3\ce{Cl2}} \underset{\text{Sodium acetate}}{\ce{H3C - \overset{\displaystyle O}{\overset{||}{C}} - O^\ominus Na^\oplus}} + \underset{\text{Chloroform}}{\ce{CHCl3}}\]