Topics
Solid State
Solid State
- Introduction to Solid State
- Amorphous and Crystalline Solids
- Crystal Lattices and Unit Cells
- Calculations Involving Unit Cell Dimensions
- Close Packed Structures of Solids
- Efficiency of Packing in Body-centred Cubic Structures
- Close Packed Structures - Formula of a Compound and Number of Voids Filled
- Number of Atoms in a Unit Cell
- Imperfections in Solids - Introduction
- Properties of Solids: Magnetic Properties
- Band Theory of Metals
- Properties of Solids: Electrical Properties
- Applications of n-type and p-type Semiconductors
Solutions and Colligative Properties
- Types of Solutions
- Expressing Concentration of Solutions
- Solubility of a Gas in a Liquid
- Solubility of a Solid in a Liquid
- Colligative Properties and Determination of Molar Mass
- Relative Lowering of Vapour Pressure
- Depression of Freezing Point
- Osmosis and Osmotic Pressure
- Abnormal Molar Masses
- Elevation of Boiling Point
Solutions
- Introduction to Solutions
- Types of Solutions
- Capacity of Solution to Dissolve Solute
- Solubility
- Vapour Pressure of Solutions of Liquids in Liquids
- Colligative Properties of Nonelectrolyte Solutions
- Vapour Pressure Lowering
- Boiling Point Elevation
- Depression in Freezing Point
- Osmosis and Osmotic Pressure
- Colligative Properties of Electrolytes
- Overview of Solutions
Ionic Equilibria
Chemical Thermodynamics and Energetic
- Chemical Thermodynamics and Energetic
- Concepts of System
- Types of Systems
- Surroundings
- Work, Heat, Energy, Extensive and Intensive Properties
- State Functions
- First Law of Thermodynamics
- Internal Energy and Enthalpy
- Hess’ Law of Constant Heat Summation
- Enthalpy of Bond Dissociation
- Combustion, Formation, Atomization, Sublimation
- Phase Transition
- Ionization and Solution
- Dilution Introduction of Entropy as a State Function
- Free Energy Change for Spontaneous and Non Spontaneous Processes
- Equilibrium Constant
- Second and Third Law of Thermodynamics
Electrochemistry
- Electrochemical Cells
- Conductance of Electrolytic Solutions
- Variation of Conductivity and Molar Conductivity with Concentration
- Electrolytic Cells and Electrolysis
- Primary Batteries
- Lead Accumulator
- Galvanic or Voltaic Cell
- Nernst Equation
- Relation Between Gibbs Energy Change and Emf of a Cell
- Fuel Cells
- Factors Affecting Corrosion
- Concept of Redox Reactions
Chemical Thermodynamics
- Chemical Thermodynamics
- Terms Used in Thermodynamics
- Nature of Heat and Work
- Expression for Pressure-volume (PV) Work
- Concept of Maximum Work
- Internal Energy (U)
- First Law of Thermodynamics
- Enthalpy (H)
- Enthalpies of Physical Transformations
- Thermochemistry
- Spontaneous (Irreversible) Process
- Overview of Chemical Thermodynamics
Chemical Kinetics
Electrochemistry
- Concept of Electrochemistry
- Electric Conduction
- Electrical Conductance of Solution
- Electrochemical Cells
- Electrolytic Cells
- Galvanic or Voltaic Cell
- Electrode Potential and Cell Potential
- Thermodynamics of Galvanic Cells
- Reference Electrodes
- Galvanic Cells Useful in Day-to-day Life
- Fuel Cells
- Electrochemical Series (Electromotive Series)
- Overview of Electrochemistry
Chemical Kinetics
- Concept of Chemical Kinetics
- Rate of Reactions
- Rate of Reaction and Reactant Concentration
- Molecularity of Reaction
- Integrated Rate Equations
- First Order Reactions
- Zero Order Reactions
- Half Life Period of a Reaction
- Pseudo First Order Reaction
- Integrated Rate Law for Gas Phase f Reactions
- Collision Theory of Bimolecular Reactions
- Temperature Dependence of Reaction Rates
- Effect of a Catalyst on the Rate of Reaction
- Overview of Chemical Kinetics
General Principles and Processes of Isolation of Elements
Elements of Groups 16, 17 and 18
- Occurrence of Elements of Groups 16, 17 and 18
- Electronic Configuration of Elements of Group 16, 17 and 18
- Atomic and Physical Properties of Elements of Group 16, 17 and 18
- Anomalous Behaviour of Oxygen
- Anomalous Behaviour of Fluorine
- Chemical Properties of Elements of Groups 16, 17 and 18
- Oxoacids
- Oxygen and Compounds of Oxygen
- Chlorine
- Group 18 Elements - The Noble gas Family
- Overview of Elements of Groups 16, 17 and 18
P-block Elements
Group 15 Elements
Group 16 Elements
Group 17 Elements
Group 18 Elements
D and F Block Elements
Transition and Inner Transition Elements
- Transition and Inner Transition Elements
- Position in the Periodic Table - d-block Elements
- Electronic Configuration of Transition and Inner Transition Elements
- Oxidation States of First Transition Series
- Physical Properties of First Transition Series
- Trends in Atomic Properties of the First Transition Series
- Preparation of Potassium Permanganate
- Chemical Properties of KMnO4
- Uses of KMnO4
- K2Cr2O7: Preparation of Potassium Dichromate
- Chemical Properties of K2Cr2O7
- Common Properties of d Block Elements
- Basic Principles of Metallurgy > Extraction of Metals
- Inner Transition (f-block) Elements: Lanthanoids and Actinoids
- Properties of f-block Elements
- Properties of Lanthanoids
- Applications of Lanthanoids
- The Actinoids
- Properties of Actinoids
- Applications of Actinoids
- Postactinoid Elements
- Overview of Transition and Inner Transition Elements
D-block Elements
- General Indroduction
- Position in the Periodic Table - d-block Elements
- Electronic Configurations of the d-block Elements
- Physical Properties of the Transition Elements (d-block)
- Some Important Compounds of Transition Elements
F-block Elements
Coordination Compounds
- Introduction
- Importance of Coordination Compounds
- Important Terms Pertaining to Coordination Compounds
- Colour in Coordination Compounds
- Magnetic Properties of Coordination Compounds
- Bonding in Coordination Compounds
- Werner’s Theory of Coordination Compounds
- Valence Bond Theory (VBT)
- Crystal Field Theory (CFT)
- Stereoisomerism
- Structural Isomerism
- Importance of Coordination Compounds
Coordination Compounds
- Concept of Coordination Compounds
- Types of Ligands
- Terms Used in Coordination Chemistry
- Classification of Complexes
- Nomenclature of Coordination Compounds
- Effective Atomic Number (EAN) Rule
- Isomerism in Coordination Compounds
- Stability of the Coordination Compounds
- Theories of Bonding in Complexes
- Importance and Applications of Coordination Compounds
- Overview of Coordination Compounds
Halogen Derivatives of Alkanes (And Arenes)
Halogen Derivatives
- Classification of Halogen Derivatives
- Nomenclature of Halogen Derivatives
- Methods of Preparation of Alkyl Halides
- Physical Properties of Halogen Derivatives
- Optical Isomerism in Halogen Derivatives
- Chemical Properties of Halogen Derivatives
- Reaction with Active Metals
- Polyhalogen Compounds
- Nomenclature of Haloalkanes and Haloarenes
- Reactions of Haloalkanes - Elimination Reactions
- Overview of Halogen Derivatives
Haloalkanes
- Concept of Haloalkanes and Haloarenes
- Nomenclature of Haloalkanes and Haloarenes
- Nature of C-X Bond of Haloalkanes and Haloarenes
- Physical Properties of Haloalkanes and Haloarenes
- Methods of Preparation of Haloalkanes
- Reactions of Haloalkanes - Nucleophilic Substitution Reactions
- R-s and D-l Configuration
Haloarenes
- Concept of Haloalkanes and Haloarenes
- Nature of C-X Bond of Haloalkanes and Haloarenes
- Reactions of Haloarenes - Nucleophilic Substitution
- Nucleophilic Substitution
- Electrophilic Substitution Reactions
- Reaction with Metals
- R-S and D-l Configurations
- Polyhalogen Compounds
Alcohols, Phenols and Ethers
Alcohols, Phenols and Ethers Alcohols
Alcohols
Phenols
- Phenols
- Methods of Preparation
- Physical and Chemical Properties
- Acidic Nature of Phenol
- Electrophillic Substitution Reactions
- Uses of Phenols
Ethers
- Ethers
- Physical and Chemical Properties of ether
- Uses of Ethers
Aldehydes, Ketones and Carboxylic Acids
- Concepts of Aldehydes, Ketones, and Carboxylic Acids
- Classification of Aldehydes, Ketones and Carboxylic Acids
- Nomenclature of Aldehydes, Ketones and Carboxylic Acids
- Preparation of Aldehydes and Ketones
- Methods of Preparation of Carboxylic Acids
- Physical Properties
- Polarity of Carbonyl Group
- Chemical Properties of Aldehydes and Ketones
- Chemical Properties of Carboxylic Acids
- Chemical Reactions of Aldehydes and Ketones - Reactions Due to α-hydrogen
- Overview: Aldehydes, Ketones and Carboxylic Acids
- Overview of Aldehydes, Ketones and Carboxylic Acids
Aldehydes, Ketones and Carboxylic Acids
Aldehydes and Ketones
- Nomenclature of Aldehydes and Ketones
- Nature of Carbonyl Group
- Physical Properties of Aldehydes and Ketones
- Chemical Reactions of Aldehydes and Ketones - Nucleophilic Addition Reactions
- Reactivity of Alpha Hydrogen in Aldehydes
- Uses of Aldehydes and Ketones
- Chemical Reactions of Aldehydes and Ketones - Other Reactions
Carboxylic Acids
- Carboxylic Acids
- Nomenclature of Carboxylic Acids
- Structure of the Carboxyl group
- Physical Properties of Carboxylic Acids
- Chemical Properties and Reactions of Carboxylic Acid
- Chemical Reactions of Carboxylic Acids - Reactions Involving Cleavege of O-H Bond
- Chemical Reactions of Carboxylic Acids - Reactions Involving Cleavege of C-OH Bond
- Chemical Reactions of Carboxylic Acids - Reactions Involving –COOH Group
- Chemical Reactions of Carboxylic Acids - Substitution Reactions in the Hydrocarbon Part
- Uses of Carboxylic Acids
Amines
Organic Compounds Containing Nitrogen
Amines
- Concept of Amines
- Classification of Amines
- Structure of Amines
- Physical Properties of Amines
- Uses of Amines
- Identification of Primary, Secondary and Tertiary Amines
- Secondary and Tertiary Amines
- Chemical Reactions of Amines - Carbylamine Reaction
- Chemical Reactions of Amines - Reaction with Nitrous Acid
- Chemical Reactions of Amines - Reaction with Arylsulphonyl Chloride
- Chemical Reactions of Amines - Electrophilic Substitution
- Preparation of Amines
Cyanides and Isocyanides
Diazonium Salts
Biomolecules
- Principal Molecules of the Living World
- Biomolecules in the Cell > Carbohydrates
- Biomolecules in the Cell > Proteins
- Overview of Biomolecules
- Overview: Biomolecules
Biomolecules
Carbohydrates
Proteins
Vitamins
- Biomolecules in the Cell > Proteins
- Classification of Vitamins
- Important Vitamins, Their Sources and Their Deficiency Diseases
Nucleic Acids
Introduction to Polymer Chemistry
- Polymer Chemistry
- Classification of Polymers> Based On Type of Different Monomers
- Some Important Polymers
- Molecular Mass and Degree of Polymerization of Polymers
- Classification of Polymers> Based on Biodegradability
- Commercially Important Polymers
- Preparation of Polytetrafluoroethene (Teflon)
- Overview of Introduction to Polymer Chemistry
Polymers
- Introduction to Polymers
- Classification of Polymers> Based on Source or Origin
- Types of Polymerisation Reactions - Addition Polymerisation or Chain Growth Polymerisation
- Types of Polymerisation Reactions - Copolymerisation
- Some Important Polymers
- Types of Polymerisation Reactions - Condensation Polymerisation Or Step Growth Polymerisation
- Nylon 6
- Nylon 66
- Types of Polymerisation Reactions - Rubber
- Classification of Polymers> Based on Biodegradability
- Preparation of Polytetrafluoroethene (Teflon)
- Preparation of Polyacrylonitrile
Green Chemistry and Nanochemistry
- Green Chemistry and Nanochemistry
- Principles of Green Chemistry
- The Role of Green Chemistry
- Introduction to Nanochemistry
- Characteristic Features of Nanoparticles
- Synthesis of Nanomaterials
- History of Nanotechnology
- Applications of Nanomaterials
- Nanoparticles and Nanotechnology
- Overview of Green Chemistry and Nanochemistry
Chemistry in Everyday Life
Chemicals in Medicines
Chemicals in Food
Cleansing Agents
Estimated time: 7 minutes
Maharashtra State Board: Class 12
Definition: Crystal Lattice
The three-dimensional arrangement of constituent particles of a crystalline solid in space, in which each particle is depicted as a point, is known as a crystal lattice.
Maharashtra State Board: Class 12
Definition: Unit cell
A unit cell is the smallest group of lattice points that, when repeated in all directions, will develop the entire lattice.
Maharashtra State Board: Class 12
Key Points: Crystal Structure
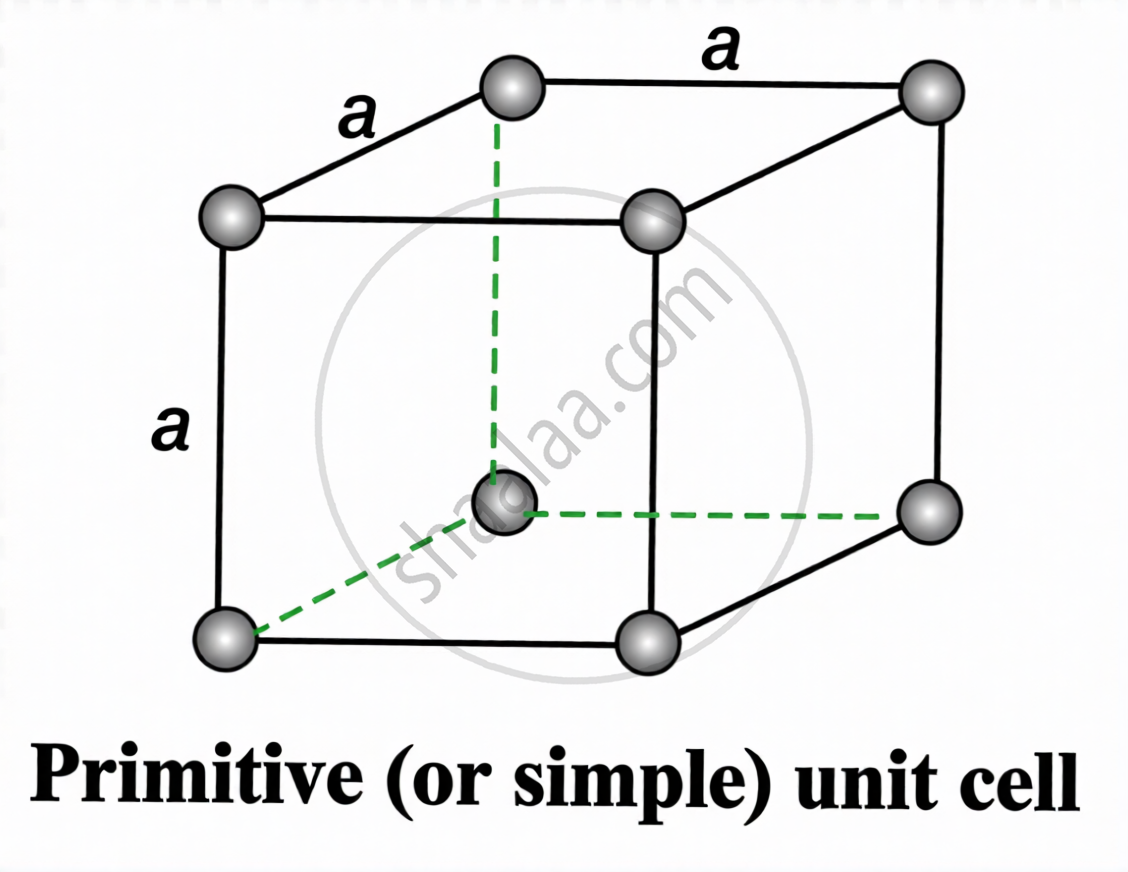
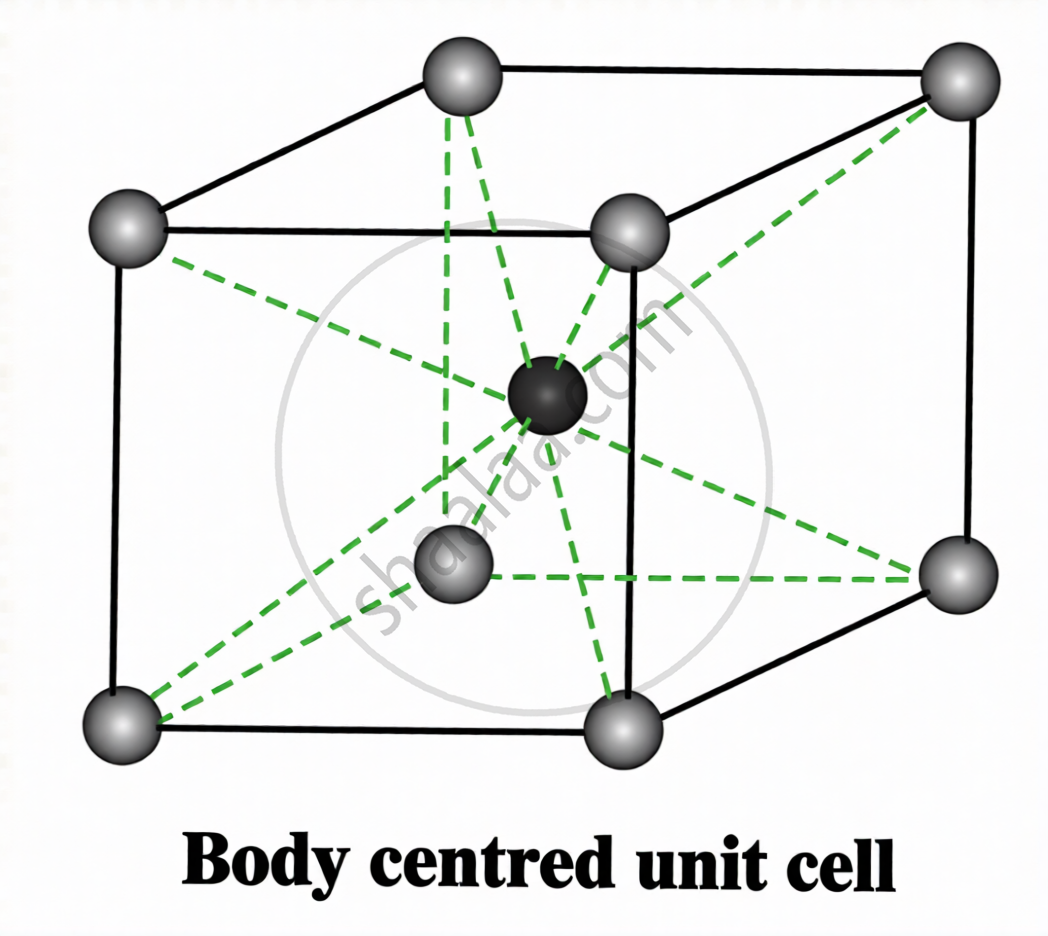
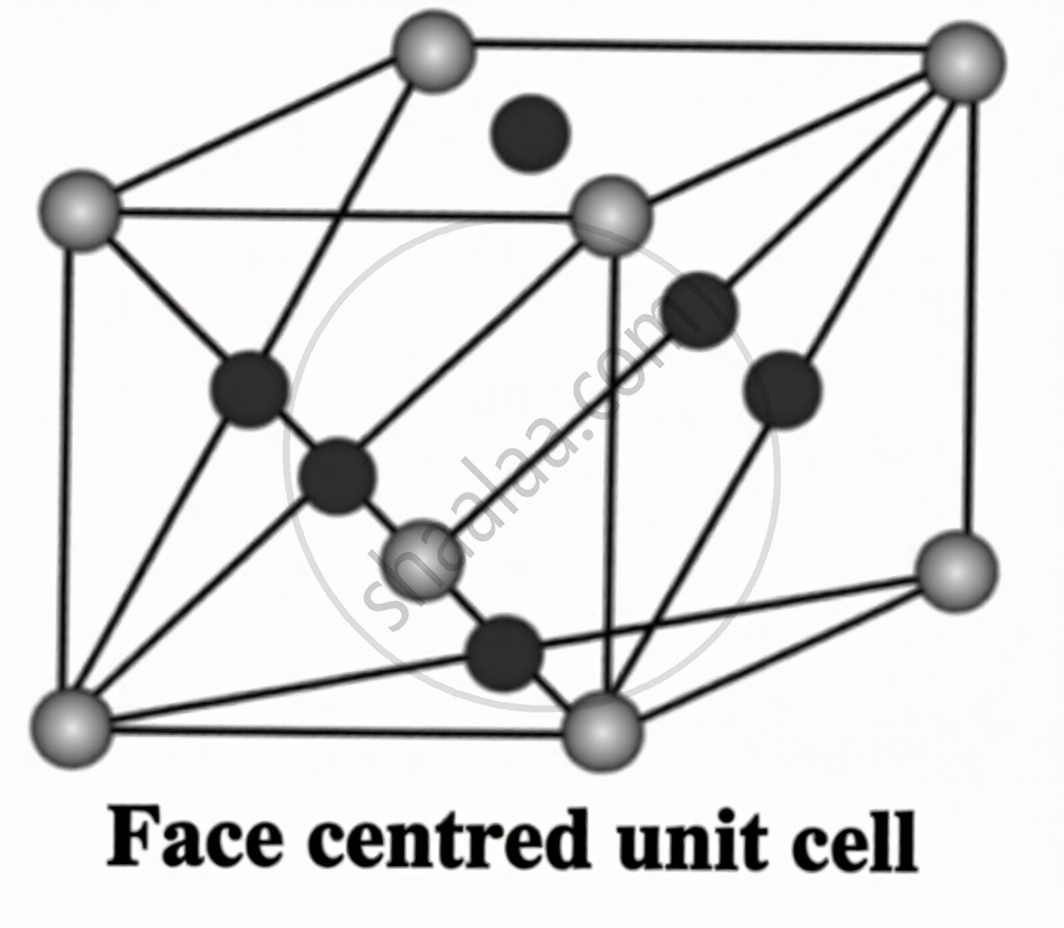
Types of Unit Cells:
| Type | Location of Particles | Atoms per Unit Cell |
|---|---|---|
|
Simple / Primitive
|
Corners only | (1/8 × 8) = 1 |
|
Body-centred (bcc)
|
Corners + 1 at body centre | (1/8 × 8) + 1 = 2 |
|
Face-centred (fcc)
|
Corners + 1 at each face centre | (1/8 × 8) + (1/2 × 6) = 4 |
| Base-centred | Corners + centres of two opposite faces | (1/8 × 8) + (1/2 × 2) = 2 |
Seven Crystal Systems:
| System | Intercepts | Angles | Bravais Lattices |
|---|---|---|---|
| Cubic | a = b = c | α = β = γ = 90° | Primitive, FCC, BCC (3) |
| Tetragonal | a = b ≠ c | α = β = γ = 90° | Primitive, BCC (2) |
| Orthorhombic | a ≠ b ≠ c | α = β = γ = 90° | Primitive, FCC, BCC, End-centred (4) |
| Monoclinic | a ≠ b ≠ c | α = γ = 90°, β ≠ 90° | Primitive, End-centred (2) |
| Triclinic | a ≠ b ≠ c | α ≠ β ≠ γ ≠ 90° | Primitive (1) |
| Hexagonal | a = b ≠ c | α = β = 90°, γ = 120° | Primitive (1) |
| Rhombohedral | a = b = c | α = β = γ ≠ 90° | Primitive (1) |