Advertisements
Chapters
▶ 2: Is Matter Around Us Pure
3: Atoms and Molecules
4: Structure of the Atom
5: The Fundamental Unit of Life
6: Tissues
7: Diversity In Living Organisms
8: Motion
9: Force and Laws of Motion
10: Gravitation
11: Work and Energy
12: Sound
13: Why Do We Fall Ill
14: Natural Resources
15: Improvement In Food Resources
![NCERT Exemplar solutions for Science [English] Class 9 chapter 2 - Is Matter Around Us Pure NCERT Exemplar solutions for Science [English] Class 9 chapter 2 - Is Matter Around Us Pure - Shaalaa.com](/images/science-english-class-9_6:5f2b1b2038084cf381bfa42c826a928c.jpg)
Advertisements
Solutions for Chapter 2: Is Matter Around Us Pure
Below listed, you can find solutions for Chapter 2 of CBSE NCERT Exemplar for Science [English] Class 9.
NCERT Exemplar solutions for Science [English] Class 9 2 Is Matter Around Us Pure Multiple Choice Questions [Pages 12 - 13]
Which of the following statements are true for pure substances?
(i) Pure substances contain only one kind of particles
(ii) Pure substances may be compounds or mixtures
(iii) Pure substances have the same composition throughout
(iv) Pure substances can be exemplified by all elements other than nickel
(i) and (ii)
(i) and (iii)
(iii) and (iv)
(ii) and (iii)
Rusting of an article made up of iron is called
corrosion and it is a physical as well as chemical change
dissolution and it is a physical change
corrosion and it is a chemical change
dissolution and it is a chemical change
A mixture of sulphur and carbon disulphide is :
heterogeneous and shows Tyndall effect
homogeneous and shows Tyndall effect
heterogeneous and does not show Tyndall effect
homogeneous and does not show Tyndall effect
Tincture of iodine has antiseptic properties. This solution is made by dissolving :
iodine in potassium iodide
iodine in acetone
iodine in water
iodine in alcohol
Which of the following are homogeneous in nature?
(i) ice
(ii) wood
(iii) soil
(iv) air
(i) and (iii)
(ii) and (iv)
(i) and (iv)
(iii) and (iv)
Which of the following are physical changes?
(i) Melting of iron metal
(ii) Rusting of iron
(iii) Bending of an iron rod
(iv) Drawing a wire of iron metal
(i), (ii) and (iii)
(i), (ii) and (iv)
(i), (iii) and (iv)
(ii), (iii) and (iv)
Which of the following are chemical changes?
(i) Decaying of wood
(ii) Burning of wood
(iii) Sawing of wood
(iv) Hammering of a nail into a piece of wood
(i) and (ii)
(ii) and (iii)
(iii) and (iv)
(i) and (iv)
Two substances, A and B were made to react to form a third substance, A2B according to the following reaction
\[\ce{2A + B -> A2 B}\]
Which of the following statements concerning this reaction are incorrect?
(i) The product A2B shows the properties of substances A and B
(ii) The product will always have a fixed composition
(iii) The product so formed cannot be classified as a compound
(iv) The product so formed is an element
(i), (ii) and (iii),
(ii), (iii) and (iv)
(i), (iii) and (iv)
(ii), (iii) and (iv)
Two chemical species X and Y combine together to form a product P which contains both X and Y
\[\ce{X + Y -> P}\]
X and Y cannot be broken down into simpler substances by simple chemical reactions. Which of the following concerning the species X, Y and P are correct?
(i) P is a compound
(ii) X and Y are compounds
(iii) X and Y are elements
(iv) P has a fixed composition
(i), (ii) and (iii)
(i), (ii) and (iv)
(ii), (iii) and (iv)
(i), (iii) and (iv)
NCERT Exemplar solutions for Science [English] Class 9 2 Is Matter Around Us Pure Short Answer Questions [Pages 14 - 16]
Suggest separation technique(s) one would need to employ to separate the following mixture.
Mercury and water
Suggest separation technique(s) one would need to employ to separate the following mixture.
Potassium chloride and ammonium chloride
Suggest separation technique(s) one would need to employ to separate the following mixture.
Common salt, water and sand
Suggest separation technique(s) one would need to employ to separate the following mixture.
Kerosene oil, water and salt
Which of the tubes in Fig. 2.1 (a) and (b) will be more effective as a condenser in the distillation apparatus?

Salt can be recovered from its solution by evaporation. Suggest some other technique for the same?
The ‘sea-water’ can be classified as a homogeneous as well as a heterogeneous mixture. Comment.
While diluting a solution of salt in water, a student by mistake added acetone (boiling point 56°C). What technique can be employed to get back the acetone? Justify your choice.
What would you observe when
a saturated solution of potassium chloride prepared at 60°C is allowed to cool to room temperature.
What would you observe when
an aqueous sugar solution is heated to dryness.
What would you observe when
a mixture of iron filings and sulphur powder is heated strongly
Explain why particles of a colloidal solution do not settle down when left undisturbed, while in the case of a suspension they do.
Smoke and fog both are aerosols. In what way are they different?
Classify the following as physical or chemical properties
The composition of a sample of steel is 98% iron, 1.5% carbon and 0.5% other elements.
Classify the following as physical or chemical properties
Zinc dissolves in hydrochloric acid with the evolution of hydrogen gas.
Classify the following as physical or chemical properties
Metallic sodium is soft enough to be cut with a knife.
Classify the following as physical or chemical properties
Most metal oxides form alkalis on interacting with water.
The teacher instructed three students ‘A’, ‘B’ and ‘C’ respectively to prepare a 50% (mass by volume) solution of sodium hydroxide (NaOH). ‘A’ dissolved 50g of NaOH in 100 mL of water, ‘B’ dissolved 50g of NaOH in 100g of water while ‘C’ dissolved 50g of NaOH in water to make 100 mL of solution. Which one of them has made the desired solution and why?
Name the process associated with the following
Dry ice is kept at room temperature and at one atmospheric pressure.
Name the process associated with the following
A drop of ink placed on the surface of water contained in a glass spreads throughout the water.
Name the process associated with the following
A potassium permanganate crystal is in a beaker and water is poured into the beaker with stirring.
Name the process associated with the following
An acetone bottle is left open and the bottle becomes empty.
Name the process associated with the following
Milk is churned to separate cream from it.
Name the process associated with the following
Settling of sand when a mixture of sand and water is left undisturbed for some time.
Name the process associated with the following
A fine beam of light entering through a small hole in a dark room illuminates the particles in its paths.
You are given two samples of water labelled as ‘A’ and ‘B’. Sample ‘A’ boils at 100°C and sample ‘B’ boils at 102°C. Which sample of the water will not freeze at 0°C? Comment.
What are the favourable qualities given to gold when it is alloyed with copper or silver for the purpose of making ornaments?
An element is sonorous and highly ductile. Under which category would you classify this element? What other characteristics do you expect the element to possess?
Give an example of each mixture having the following characteristics. Suggest a suitable method to separate the components of this mixture
A volatile and a non-volatile component.
Give an example of each mixture having the following characteristics. Suggest a suitable method to separate the components of this mixture
Two volatile components with appreciable differences in boiling points.
Give an example of each mixture having the following characteristics. Suggest a suitable method to separate the components of this mixture
Two immiscible liquids.
Give an example of each mixture having the following characteristics. Suggest a suitable method to separate the components of this mixture
One of the components changes directly from solid to gaseous state.
Give an example of each mixture having the following characteristics. Suggest a suitable method to separate the components of this mixture
Two or more coloured constituents are soluble in some solvent.
Fill in the blanks
A colloid is a ______ mixture and its components can be separated by the technique known as ______.
Ice, water and water vapour look different and display different ______ properties but they are ______ the same.
A mixture of chloroform and water are taken in a separating funnel is mixed and left undisturbed for some time. The upper layer in the separating funnel will be of ______ and the lower layer will be that of ______.
A mixture of two or more miscible liquids, for which the difference in the boiling points is less than 25 K can be separated by the process called ______.
When light is passed through water containing a few drops of milk, it shows a bluish tinge. This is due to the ______ of light by milk and the phenomenon is called ______. This indicates that milk is a ______ solution.
Sucrose (sugar) crystals obtained from sugarcane and beetroot are mixed together. Will it be a pure substance or a mixture? Give reasons for the same.
Give some examples of the Tyndall effect observed in your surroundings?
Can we separate alcohol dissolved in water by using a separating funnel? If yes, then describe the procedure. If not, explain.
On heating calcium carbonate gets converted into calcium oxide and carbon dioxide.
(a) Is this a physical or a chemical change?
(b) Can you prepare one acidic and one basic solution by using the products formed in the above process? If so, write the chemical equation involved.
Non-metals are usually poor conductors of heat and electricity. They are non-lustrous, non-sonorous, non-malleable and are coloured.
Name a lustrous non-metal.
Non-metals are usually poor conductors of heat and electricity. They are non-lustrous, non-sonorous, non-malleable and are coloured.
Name a non-metal which exists as a liquid at room temperature.
Non-metals are usually poor conductors of heat and electricity. They are non-lustrous, non-sonorous, non-malleable and are coloured.
The allotropic form of a non-metal is a good conductor of electricity. Name the allotrope.
Non-metals are usually poor conductors of heat and electricity. They are non-lustrous, non-sonorous, non-malleable and are coloured.
Name a non-metal which is known to form the largest number of compounds.
Non-metals are usually poor conductors of heat and electricity. They are non-lustrous, non-sonorous, non-malleable and are coloured.
Name a non-metal other than carbon that shows allotropy.
Non-metals are usually poor conductors of heat and electricity. They are non-lustrous, non-sonorous, non-malleable and are coloured.
Name a non-metal which is required for combustion.
Classify the substances given in Fig. 2.2 into elements and compounds

Which of the following are not compounds?
- Chlorine gas
- Potassium chloride
- Iron
- Iron sulphide
- Aluminium
- Iodine
- Carbon
- Carbon monoxide
- Sulphur powder
NCERT Exemplar solutions for Science [English] Class 9 2 Is Matter Around Us Pure Long Answer Questions [Pages 17 - 18]
Fractional distillation is suitable for the separation of miscible liquids with a boiling point difference of about 25 K or less. What part of the fractional distillation apparatus makes it efficient and possess an advantage over a simple distillation process. Explain using a diagram.
Under which category of mixtures will you classify alloys and why?
A solution is always a liquid. Comment.
Can a solution be heterogeneous?
Iron filings and sulphur were mixed together and divided into two parts, ‘A’ and ‘B’. Part ‘A’ was heated strongly while Part ‘B’ was not heated. Dilute hydrochloric acid was added to both Parts and evolution of gas was seen in both cases. How will you identify the gases that evolved?
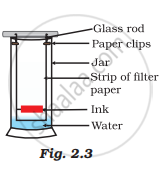
A child wanted to separate the mixture of dyes constituting a sample of ink. He marked a line by the ink on the filter paper and placed the filter paper in a glass containing water as shown in Fig.2.3. The filter paper was removed when the water moved near the top of the filter paper.
(i) What would you expect to see, if the ink contains three different coloured components?
(ii) Name the technique used by the child.
(iii) Suggest one more application of this technique.
A group of students took an old shoebox and covered it with black paper from all sides. They fixed a source of light (a torch) at one end of the box by making a hole in it and making another hole on the other side to view the light. They placed a milk sample contained in a beaker/tumbler in the box as shown in Fig.2.4. They were amazed to see that milk taken in the tumbler was illuminated. They tried the same activity by taking a salt solution but found that light simply passed through it?
(a) Explain why the milk sample was illuminated. Name the phenomenon involved.
(b) The same results were not observed with a salt solution. Explain.
(c) Can you suggest two more solutions that would show the same effect as shown by the milk solution?

Classify each of the following, as a physical or a chemical change. Give reasons.
(a) Drying of a shirt in the sun.
(b) Rising of hot air over a radiator.
(c) Burning of kerosene in a lantern.
(d) Change the colour of black tea by adding lemon juice to it.
(e) Churning of milk cream to get butter.
During an experiment, the students were asked to prepare a 10% (Mass/Mass) solution of sugar in water. Ramesh dissolved 10g of sugar in 100g of water while Sarika prepared it by dissolving 10g of sugar in water to make 100g of the solution.
(a) Are the two solutions of the same concentration
(b) Compare the mass % of the two solutions.
You are provided with a mixture containing sand, iron filings, ammonium chloride and sodium chloride. Describe the procedures you would use to separate these constituents from the mixture?
Arun has prepared a 0.01% (by mass) solution of sodium chloride in water. Which of the following correctly represents the composition of the solutions?
1.00 g of NaCl + 100g of water
0.11g of NaCl + 100g of water
0.0l g of NaCl + 99.99g of water
0.10 g of NaCl + 99.90g of water
Calculate the mass of sodium sulphate required to prepare its 20% (mass percent) solution in 100g of water?
Solutions for 2: Is Matter Around Us Pure
![NCERT Exemplar solutions for Science [English] Class 9 chapter 2 - Is Matter Around Us Pure NCERT Exemplar solutions for Science [English] Class 9 chapter 2 - Is Matter Around Us Pure - Shaalaa.com](/images/science-english-class-9_6:5f2b1b2038084cf381bfa42c826a928c.jpg)
NCERT Exemplar solutions for Science [English] Class 9 chapter 2 - Is Matter Around Us Pure
Shaalaa.com has the CBSE Mathematics Science [English] Class 9 CBSE solutions in a manner that help students grasp basic concepts better and faster. The detailed, step-by-step solutions will help you understand the concepts better and clarify any confusion. NCERT Exemplar solutions for Mathematics Science [English] Class 9 CBSE 2 (Is Matter Around Us Pure) include all questions with answers and detailed explanations. This will clear students' doubts about questions and improve their application skills while preparing for board exams.
Further, we at Shaalaa.com provide such solutions so students can prepare for written exams. NCERT Exemplar textbook solutions can be a core help for self-study and provide excellent self-help guidance for students.
Concepts covered in Science [English] Class 9 chapter 2 Is Matter Around Us Pure are Mixture, Introduction to Solutions, Matter (Substance), Concentration of a Solution, Suspension Solution, Colloidal Solution, Evaporation Method, Compound, Types of Mixtures, Solvent Extraction (Using a Separating Funnel Method), Chromatography Method, Simple Distillation Method, Fractional Distillation Method, Crystallisation Method, Classification of Change: Physical Changes, Chemical Reaction, Pure Substances, Elements, Sublimation, Natural substances.
Using NCERT Exemplar Science [English] Class 9 solutions Is Matter Around Us Pure exercise by students is an easy way to prepare for the exams, as they involve solutions arranged chapter-wise and also page-wise. The questions involved in NCERT Exemplar Solutions are essential questions that can be asked in the final exam. Maximum CBSE Science [English] Class 9 students prefer NCERT Exemplar Textbook Solutions to score more in exams.
Get the free view of Chapter 2, Is Matter Around Us Pure Science [English] Class 9 additional questions for Mathematics Science [English] Class 9 CBSE, and you can use Shaalaa.com to keep it handy for your exam preparation.
