Advertisements
Advertisements
If a gas is compressed isothermally then the r.m.s. velocity of the molecules ______.
Concept: undefined >> undefined
If 'ΔQ' is the amount of heat supplied to 'n' moles of a diatomic gas at constant pressure, 'Δ U' is the change in internal energy and 'Δ W' is the work done, then Δ W : Δ U : Δ Q is ______.
Concept: undefined >> undefined
Advertisements
An ideal gas at 27°C is compressed adiabatically to (8/27) of its original volume. If ratio of specific heats, y = 5/3 then the rise in temperature of the gas is ______.
Concept: undefined >> undefined
A glass plate is partly dipped vertically in the mercury and the angle of contact is measured. If the plate is inclined, then the angle of contact will ____________.
Concept: undefined >> undefined
A water barrel stands on a table of height 'h'. A small hole is made on the wall of barrel at its bottom. If the stream of water coming out of the hole strikes the ground at horizontal distance 'R' from the table, the depth 'd' of water in the barrel is ______.
Concept: undefined >> undefined
A particle is moving with speed v = b`sqrtx` along positive x-axis. Calculate the speed of the particle at time t = τ (assume that the particle is at origin at t = 0).
Concept: undefined >> undefined
A monoatomic gas at pressure P and volume V is suddenly compressed to one eights of its original volume. The final pressure at constant entropy will be ______.
Concept: undefined >> undefined
For an ideal gas graph is shown for three processes. Process 1, 2 and 3 are respectively.
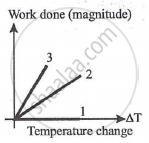
Concept: undefined >> undefined
With the increase in temperature, the angle of contact ______.
Concept: undefined >> undefined
A prism has a refracting angle of 60°. When placed in the position of minimum deviation, it produces a deviation of 30°. The angle of incidence is ______.
Concept: undefined >> undefined
A ray of light travelling through rarer medium is incident at very small angle i on a glass slab and after refraction its velocity is reduced by 20%. The angle of deviation is ______.
Concept: undefined >> undefined
In a thermodynamic system, W represents the work done by the system and ΔU is the increase in internal energy. Which of the following statements is true?
Concept: undefined >> undefined
Which of the following statements is correct regarding the angle of contact θc of a liquid drop with a flat solid surface?
Concept: undefined >> undefined
A parallel plate capacitor is charged to 100 × 10-6 C. Due to radiations, falling from a radiating source, the plate loses charge at the rate of 2 × 10-7 Cs-1. The magnitude of displacement current is ______.
Concept: undefined >> undefined
In an adiabatic expansion, a gas does 25 J of work, while in an adiabatic compression 100 J of work is done on a gas. The change of internal energy in the two processes respectively, are ______.
Concept: undefined >> undefined
Calculate the work done (WAB ) by the gas, if 5 moles of an ideal gas is carried by a quasi state isothermal process at 500 K to twice its volume ______.
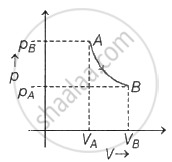
Concept: undefined >> undefined
540 g of ice at 0°C is mixed with 540 g of water at 80°C. The final temperature of the mixture will be ______.
Concept: undefined >> undefined
If in an adiabatic process, the pressure is increased by 2/3%, then the volume decreases by ______.
`("Assume", "C"_"p"/"C"_"v"=3/2)`
Concept: undefined >> undefined
A cycle tyre bursts suddenly, this represents an ______.
Concept: undefined >> undefined
The ratio of energy required to raise a satellite of mass 'm' to a height 'h' above the earth's surface of that required to put it into the orbit at same height is ______.
[R = radius of the earth]
Concept: undefined >> undefined
