Advertisements
Advertisements
Question
For an ideal gas graph is shown for three processes. Process 1, 2 and 3 are respectively.
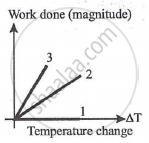
Options
Isobaric, adiabatic, isochoric
Adiabatic, isobaric, isochoric
Isochoric, adiabatic, isobaric
Isochoric, isobaric, adiabatic
MCQ
Advertisements
Solution
Isochoric, isobaric, adiabatic
Explanation:
Isochoric processes dV = 0
W = 0 processes 1
Isobaric: W = PΔV = nRΔT
Adiabatic |W| = `("nR"Delta"T")/(gamma-1)` 0 < γ - 1 < 1
As work is done more in the case of adiabatic processes, process 3 is adiabatic and process 2 is isobaric.
shaalaa.com
Is there an error in this question or solution?
