Advertisements
Advertisements
Complete the following:
\[\ce{CH3CN ->[1. AlH(i - Bu)2][2. H2O] 'A' ->[H2N-OH][H^+] 'B'}\]
Concept: undefined >> undefined
Write IUPAC name of the following compound:
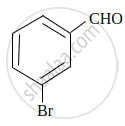
Concept: undefined >> undefined
Advertisements
Read the passage given below and answer the questions that follow:
|
Oxidation-reduction reactions are commonly known as redox reactions. They involve transfer of electrons from one species to another. In a spontaneous reaction, energy is released which can be used to do useful work. The reaction is split into two half-reactions. Two different containers are used and a wire is used to drive the electrons from one side to the other and a Voltaic/Galvanic cell is created. It is an electrochemical cell that uses spontaneous redox reactions to generate electricity. A salt bridge also connects to the half-cells. The reading of the voltmeter gives the cell voltage or cell potential or electromotive force. If \[\ce{E^0_{cell}}\] is positive the reaction is spontaneous and if it is negative the reaction is non-spontaneous and is referred to as electrolytic cell. Electrolysis refers to the decomposition of a substance by an electric current. One mole of electric charge when passed through a cell will discharge half a mole of a divalent metal ion such as Cu2+. This was first formulated by Faraday in the form of laws of electrolysis.
|
- Is silver plate the anode or cathode? (1)
- What will happen if the salt bridge is removed? (1)
- When does electrochemical cell behaves like an electrolytic cell? (1)
- (i) What will happen to the concentration of Zn2+ and Ag+ when Ecell = 0. (1)
(ii) Why does conductivity of a solution decreases with dilution? (1)
OR
The molar conductivity of a 1.5 M solution of an electrolyte is found to be 138.9 S cm2mol-1. Calculate the conductivity of this solution. (2)
Concept: undefined >> undefined
Write the IUPAC name of the following complex:
[Pt(NH3)6]Cl4
Concept: undefined >> undefined
Using IUPAC norms, write the formula for the following:
Pentaamminenitrito-N-Cobalt (III)
Concept: undefined >> undefined
Using IUPAC norms write the formula for the following:
Tetrahydroxidozincate (II)
Concept: undefined >> undefined
Differentiate between the following:
Adsorption and Absorption
Concept: undefined >> undefined
The following questions are case-based questions. Read the passage carefully and answer the questions that follow:
| Carbohydrates are optically active polyhydroxy aldehydes and ketones. They are also called saccharides. All those carbohydrates which reduce Fehling's solution and Tollen's reagent are referred to as reducing sugars. Glucose, the most important source of energy for mammals, is obtained by the hydrolysis of starch. Vitamins are accessory food factors required in the diet. Proteins are the polymers of α-amino acids and perform various structural and dynamic functions in the organisms. Deficiency of vitamins leads to many diseases. |
Answer the following:
(a) The penta-acetate of glucose does not react with Hydroxylamine. What does it indicate? (1)
(b) Why cannot vitamin C be stored in our body? (1)
(c) Define the following as related to proteins: (2)
- Peptide linkage
- Denaturation
OR
(c) Define the following as related to carbohydrates: (2)
- Anomers
- Glycosidic linkage
Concept: undefined >> undefined
Which of the following belongs to the class of vinyl halides?
Concept: undefined >> undefined
What is the secondary valency of cobalt in [Co(en2)Cl2]+?
Concept: undefined >> undefined
What should be the signs (positive/negative) for \[\ce{E^0_{cell}}\] and ΔG0 for a spontaneous redox reaction occurring under standard conditions?
Concept: undefined >> undefined
Assertion (A): [Co(NH3)5SO4]Cl gives a white precipitate with silver nitrate solution.
Reason (R): The complex dissociates to give Cl− and \[\ce{SO^{2-}_4}\] ions.
Concept: undefined >> undefined
What is IUPAC name of the ketone A, which undergoes iodo form reaction to give \[\ce{CH3CH = C(CH3)COONa}\] and yellow precipitate of \[\ce{CH3}\]?
Concept: undefined >> undefined
Convert the following:
Bromobenzene to benzoic acid
Concept: undefined >> undefined
Calculate the λ0m for Cl- ion from the data given below:
∧0m MgCl2 = 258.6 Scm2 mol-1 and λ0m Mg2+ = 106 Scm2 mol-1
Concept: undefined >> undefined
Name the following halides according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3CH2C(CH3)2CH2I}\]
Concept: undefined >> undefined
Name the following halide according to IUPAC system and classify it as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
\[\ce{CH2CH2C(CH3)2CH2I}\]
Concept: undefined >> undefined
Write the structure of the following compound:
Di-sec-butyl ketone
Concept: undefined >> undefined
Write the structure of the following compound:
Di-sec-butyl ketone
Concept: undefined >> undefined
Name the following halide according to IUPAC system and classify it as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
\[\ce{CH3C(C2H5)2CH2Br}\]
Concept: undefined >> undefined

