Advertisements
Advertisements
Complete the reaction with the main product formed:

Concept: undefined >> undefined
Convert bromoethane to propanamine.
Concept: undefined >> undefined
Advertisements
Give the mechanism of the following reaction:
\[\ce{CH3CH2OH ->[H2SO4][413 K] CH3CH2-O-CH2CH3 + H2O}\]
Concept: undefined >> undefined
The following questions are case-based questions. Read the passage carefully and answer the questions that follow:
|
Nucleophilic Substitution: Influences of solvent polarity: The reaction rate (SN2) of 2-bromopropane and NaOH in ethanol containing 40% water is twice slower than in absolute ethanol. Hence the level of solvent polarity has an influence on both SN1 and SN2 reactions but with different results. Generally speaking, a weak polar solvent is favourable for SN2 reaction, while a strong polar solvent is favourable for SN1. Generally speaking, the substitution reaction of tertiary haloalkane is based on SN1 mechanism in solvents with a strong polarity (for example ethanol containing water). |
Answer the following questions:
(a) Why racemisation occurs in SN1? (1)
(b) Why is ethanol less polar than water? (1)
(c) Which one of, the following in each pair is more reactive towards SN2 reaction? (2)
(i) CH3 – CH2 – I or CH3CH2 – Cl
(ii)
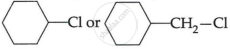
OR
(c) Arrange the following in the increasing order of their reactivity towards SN1 reactions: (2)
(i) 2-Bromo-2-methylbutane, 1-Bromo-pentane, 2-Bromo-pentane
(ii) 1-Bromo-3-methylbutane, 2-Bromo-2-methylbutane, 2-Bromo-3- methylbutane
Concept: undefined >> undefined
An organic compound 'A' with molecular formula C5H8O2 is reduced to n-pentane with hydrazine followed by heating with NaOH and glycol. 'A' forms a dioxime with hydroxylamine and gives a positive iodoform and Tollen's test. Identify 'A' and give its reaction for iodoform and Tollen's test.
Concept: undefined >> undefined
An organic compound 'A' with the molecular formula C4H8O2 undergoes acid hydrolysis to form two compounds 'B' and 'C'. Oxidation of 'C' with acidified potassium permanganate also produces 'B'. Sodium salt of 'B' on heating with soda lime gives methane.
- Identify 'A', 'B' and 'C'.
- Out of 'B' and 'C', which will have higher boiling point? Give reason.
Concept: undefined >> undefined
Hydrolysis of sucrose is called ______.
Concept: undefined >> undefined
You are given four organic compounds “A”, “B” , “C” and “D”. The compounds “A”, “B” and “C” form an orange-red precipitate with 2, 4 DNP reagent. Compounds “A” and “B” reduce Tollen’s reagent while compounds “C” and “D” do not. Both “B” and “C” give a yellow precipitate when heated with iodine in the presence of NaOH. Compound “D” gives brisk effervescence with sodium bicarbonate solution. Identify “A”, “B”, “C” and “D” given the number of carbon atoms in three of these carbon compounds is three while one has two carbon atoms. Give an explanation for our answer.
Concept: undefined >> undefined
When sucrose is hydrolysed the optical rotation values are measured using a polarimeter and are given in the following table:
| S. No. | Time (hours) | Specific Rotation |
| 1 | 0 | + 66.5° |
| 2 | ∞ | - 39.9° |
- Account for the two specific rotation values.
- What is the specific name given to sucrose based on the above observation?
- One of the products formed during the hydrolysis of sucrose is a glucose, that reacts with hydroxylamine to give compound A. Identify compound A.
Concept: undefined >> undefined
An organic compound A with the molecular formula (+) C4H9Br undergoes hydrolysis to form (+) C4H9OH. Give the structure of A and write the mechanism of the reaction.
Concept: undefined >> undefined
Acetic anhydride from acetic acid
Concept: undefined >> undefined
Explain why Grignard reagents should be prepared under anhydrous conditions.
Concept: undefined >> undefined
Which alkyl halide from the following pair would you expect to react more rapidly by an SN2 mechanism? Explain your answer.
\[\begin{array}{cc}\ce{CH3CH2CHCH3}\\
\phantom{.....}|\\
\phantom{......}\ce{Br}\\
\end{array}\] or \[\begin{array}{cc}\phantom{.......}\ce{CH3}\\
\phantom{...}|\\
\ce{H3C - C - Br}\\
\phantom{...}|\\
\phantom{.......}\ce{CH3}\\
\end{array}\]
Concept: undefined >> undefined
Which alkyl halide from the following pair would you expect to react more rapidly by an SN2 mechanism? Explain your answer.
\[\begin{array}{cc}
\ce{CH3CHCH2CH2Br}\\
|\phantom{.............}\\
\ce{CH3}\phantom{..........}\\
\end{array}\] or \[\begin{array}{cc}
\ce{CH3CH2CHCH2Br}\\
|\\
\phantom{...}\ce{CH3}\\
\end{array}\]
Concept: undefined >> undefined
Which one of the following is a food preservative?
Equanil, Morphine, Sodium benzoate
Concept: undefined >> undefined
Write the chemical equations involved in the following reactions:
Carbylamine reaction
Concept: undefined >> undefined
How do you convert:
2-bromobutane to but-2-ene
Concept: undefined >> undefined
Write the structural difference between starch and cellulose.
Concept: undefined >> undefined
[NiCl4]2− is paramagnetic, while [Ni(CO)4] is diamagnetic, though both are tetrahedral. Why? (Atomic number of Ni = 28)
Concept: undefined >> undefined
Write the main products when n-butyl chloride is treated with alcoholic KOH.
Concept: undefined >> undefined
