Advertisements
Advertisements
Question
Write the chemical formula of the following compound in a step-by-step manner –
Calcium hydroxide
Advertisements
Solution
Write the formula of
| Calcium hydroxide | Symbol | Valency |
| Calcium | \[\ce{Ca}\] | \[\ce2^+\] |
| Hydroxide | \[\ce{OH}\] | \[\ce1^-\] |
Step I – Write each symbol with its valency.
Positive ion is written first
\[\ce{Ca}^{2+}\] \[\ce{[OH]}^{1-}\]
Step II – Interchange the valencies and ignoring the positive and negative signs.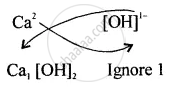
The Formula is \[\ce{Ca[OH]2}\]
APPEARS IN
RELATED QUESTIONS
Write the molecular formula of the following compounds of Iron (II) sulphide and.
Find the number and names of elements present in them and calculate their molecular masses.
Write the molecular formulae for the following compounds and name the elements present of Common salt.
The valency of aluminium is 3. Write the valency of other radicals present in the following compound.
Aluminium chloride
What is variable valency ? Give two examples of elements showing variable valency.
Write the chemical formula of the following compound in a step-by-step manner –
Potassium nitrate
Write the chemical formula of the following compound in a step-by-step manner –
Calcium bicarbonate
Write the chemical formula of the following compound in a step-by-step manner –
Sodium bisulphate
Write the chemical formula of the following compound in a step-by-step manner –
Magnesium nitride
Write the chemical formula of the following compound in a step-by-step manner –
Copper [I] sulphide
Complete the statement given below by filling in the blanks with the correct words.
The valency of iron in FeO is _______ of chlorine [chloride] in CaCl2 is ________ and of dichromate in K2 Cr2 O7 is ________.
