Advertisements
Advertisements
Question
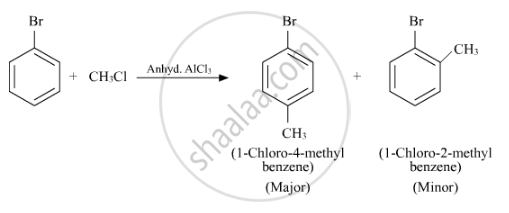
Write the structure of the major product in each of the following reaction :

Advertisements
Solution
RELATED QUESTIONS
Which would undergo SN1 reaction faster in the following pair and why?

Which compound in the following pair will react faster in SN2 reaction with OH−?
(CH3)3CCl or CH3Cl
What is the action of the following on ethyl bromide:
moist silver oxide
Optically active isomers but not mirror images are called ____________.
Which of the following is the correct order of decreasing SN2 reactivity?
Complete the following analogy:
Same molecular formula but different structures: A : : Non superimposable mirror images: B
Aryl halides are extremely less reactive towards nucleophilic substitution. Predict and explain the order of reactivity of the following compounds towards nucleophilic substitution:
| (I) |  |
| (II) |  |
| (III) |  |
Which one of the following compounds is more reactive towards SN1 reaction?
Consider the reactions,
(i) \[\begin{array}{cc}
\phantom{}\ce{\underset{}{(CH3)2CH - CH2Br} ->[C2H5OH] \underset{}{(CH3)2CH - CH2OC2H5 + HBr}}\\
\end{array}\]
(ii) \[\begin{array}{cc}
\phantom{}\ce{\underset{}{(CH3)2CH - CH2Br} ->[C2H5O-] \underset{}{(CH3)2CH - CH2OC2H5 + Br-}}\\
\end{array}\]
The mechanisms of reactions (i) and (ii) are respectively:
The correct order of increasing reactivity of
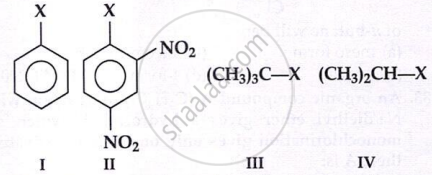
C-X bond towards nucleophile in the following compounds is:
