Advertisements
Advertisements
Question
Write the name and molecular formula of the first member of the homologous series of alkynes.
Advertisements
Solution
APPEARS IN
RELATED QUESTIONS
Write the molecular formula of the 2nd and 3rd member of the homologous series where the first member is ethyne.
What is the next higher homologue of methanol (CH3OH)?
The molecular formula of a homologue of butane is:
(a) C4H8
(b) C3H6
(c) C4H6
(d) C3H8
Give the names and the structural formula of the first three members of the homologous series of alkanes.
What is the difference in the molecular formula of any two adjacent homologues:
(i) In terms of molecular mass
(ii) In terms of number and kind of atoms per molecule?
The general molecular formula for the homologous series of alkynes is _______.
Observe the structural formula and answer the following questions.
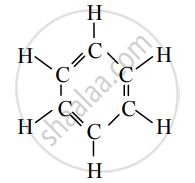
- Write the name of the given hydrocarbon.
- The given hydrocarbon is included in which type of hydrocarbon?
- What is the kind of compounds with the above characteristic structure called?
Name and draw the electron dot structure of first homologue of alkynes series.
Name the third homologue of alcohols.
Name the third homologue of aldehydes.
