Advertisements
Advertisements
Question
Write down the electronic configuration of the following
- 2713X,
- 3517Y.
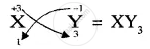
Write down the number of electrons in X and neutrons in Y and the formula of the compound formed by X and Y.[XY3]
Advertisements
Solution
Electronic configuration of:
(1) 2713X number of electrons = 13
Distribution of elecrtrons in different ordits
13 = [2, 8, 3]
K L M
Number of neutrons in Y
= A − Z = 35 − 17 = 18
(2) 3517Y number of electrons = 17
Distribution of electrons in different orbits
17 = [2, 8, 7]
K L M
Formula of compound formed is
APPEARS IN
RELATED QUESTIONS
State whether the following statement is true or false :
The cathode rays obtained from all the gases consist of negatively charged particles called electrons.
What is an electron? State its relative mass and charge.
All the gases from cathode rays and anode rays when electricity is passed through them :What does the formation of cathode rays tell us about the atoms?
How does an electron differ from a neutron?
What are inert elements?
Choose the correct option
The number of valence electrons in O2- is :
Choose the correct option:
The mass number of an atom whose unipositive ion has 10 electrons and 12 neutrons is :
What is the term defined below?
A bond formed by a shared pair of electrons, each bonding atom contributing one electron to the pair.
_______ is a negatively charged particle.
Assertion: Helium has four electrons in the outermost orbit.
Reason: Neon has eight electrons in the outermost orbit.
