Advertisements
Advertisements
प्रश्न
Write down the electronic configuration of the following
- 2713X,
- 3517Y.
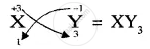
Write down the number of electrons in X and neutrons in Y and the formula of the compound formed by X and Y.[XY3]
Advertisements
उत्तर
Electronic configuration of:
(1) 2713X number of electrons = 13
Distribution of elecrtrons in different ordits
13 = [2, 8, 3]
K L M
Number of neutrons in Y
= A − Z = 35 − 17 = 18
(2) 3517Y number of electrons = 17
Distribution of electrons in different orbits
17 = [2, 8, 7]
K L M
Formula of compound formed is
APPEARS IN
संबंधित प्रश्न
Fill in the blank of the following statement :
Cathode rays are a beam of fast moving _________.
The ion of an element has 3 positive charges. The mass number of atom of this element is 27 and the number of neutrons is 14. What is the number of electrons in the ion?
Which sub-atomic particle was discovered by Chadwick.
An element A has 1 electron in its first shell. It combines with element B having 7 electrons in its third shell. What type of bond is formed?
If electrons are getting added to en element Y; then
What charge will Y migrate to during the process of electrolysis?
Give a simple diagram to show the arrangement of the electrons in an atom of sulphur.
What is the relation between the number of protons and the number of electrons in an atom?
Give a reason why noble gases have a stable electronic configuration.
_______ is a negatively charged particle.
Select the INCORRECT statement.
