Advertisements
Advertisements
प्रश्न
Write down the electronic configuration of the following
- 2713X,
- 3517Y.
Write down the number of electrons in X and neutrons in Y and the formula of the compound formed by X and Y.[XY3]
Advertisements
उत्तर
Electronic configuration of:
(1) 2713X number of electrons = 13
Distribution of elecrtrons in different ordits
13 = [2, 8, 3]
K L M
Number of neutrons in Y
= A − Z = 35 − 17 = 18
(2) 3517Y number of electrons = 17
Distribution of electrons in different orbits
17 = [2, 8, 7]
K L M
Formula of compound formed is
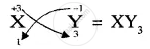
APPEARS IN
संबंधित प्रश्न
The atom of an element X has 7 electrons in its M shell.
Is it a metal or a non-metal?
Name the sub-atomic particle whose charge is -1.
Explain the rule according to which electrons are filled in various energy levels.
An atom X has three electrons more than the noble gas configuration. What type of ion will it form?
The electronic configuration of nitrogen is (2, 5). How many electrons in the outer shell of a nitrogen atom are not involved in the formation of a nitrogen molecule?
What is the term defined below?
A bond formed by a shared pair of electrons, each bonding atom contributing one electron to the pair.
What are the noble gases?
Give a reason why noble gases have a stable electronic configuration.
Atoms of all metals will have ______ electrons in their outermost orbit.
Anode rays travel in straight lines.
