Advertisements
Advertisements
Question
Write balanced equation to satisfy the following statement:
\[\ce{Acid + Active metal -> Salt + hydrogen}\]
Advertisements
Solution
\[\ce{Mg +2HCl → MgCl2 + H2}\]
APPEARS IN
RELATED QUESTIONS
Define the term ‘basicity’ of an acid.
Give two examples of the following:
oxy-acid
Name an acid used to remove ink spots.
Which acid is used for getting chloride salt?
Vinu and Priyan take their lunch at school. Vinu eats lemon rice and Priyan eats curd rice. Both lemon rice and curd rice are sour in taste. What is the reason?
Fill in the cross word given in figure with the help of the clues provided.
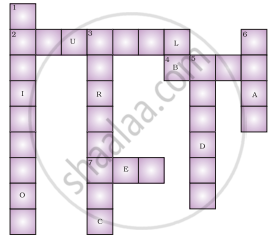
Across
(2) The solution which does not change the colour of either red or blue litmus.
(4) Phenolphthalein gives pink colour in this type of solution.
(7) Colour of blue litmus in lemon juice.
Down
(1) It is used to test whether a substance is acidic or basic.
(3) It is a natural indicator and gives pink colour in basic solution.
(5) Nature of ant’s sting.
(6) It is responsible for the increase in temperature during a neutralisation reaction.
Which is the strongest acid in the following?
You are supplied with five solutions: A, B, C, D and E with pH values as follows: A = 1.8, B = 7, C = 8.5, D = 13, and E = 5
Classify these solutions as neutral, slightly or strongly acidic and slightly or strongly alkaline.
Which solution would be most likely to liberate hydrogen with powdered zinc metal. Give a word equation for each reaction.
Select an oxide which dissolves in water forming an acid.
Draw the structure of Hydronium ion.
