Advertisements
Advertisements
Question
Write a short note on ammonolysis.
Advertisements
Solution
The carbon-halogen bond in alkyl or benzyl halides can be easily cleaved by a nucleophile. Hence, an alkyl or benzyl halide on reaction with an ethanolic solution of ammonia undergoes a nucleophilic substitution reaction in which the halogen atom is replaced by an amino (–NH2) group. This process of cleavage of the C–X bond by an ammonia molecule is known as ammonolysis. The reaction is carried out in a sealed tube at 373 K. The primary amine thus obtained behaves as a nucleophile and can further react with alkyl halide to form secondary and tertiary amines and finally quaternary ammonium salt.
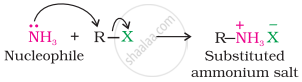
\[\ce{\underset{(1^\circ)}{RNH2} ->[RX] \underset{(2^\circ)}{R2NH} ->[RX] \underset{(3^\circ)}{R3N} ->[RX] \underset{Quaternary ammonium salt}{R4\overset{+}{N}\overset{-}{X}}}\]
The free amine can be obtained from the ammonium salt by treatment with a strong base:
\[\ce{R-\overset{+}{N}H3\overset{-}{X} + NaOH -> R-NH2 + H2O + \overset{+}{N}a\overset{-}{X}}\]
Ammonolysis has the disadvantage of yielding a mixture of primary, secondary and tertiary amines and also a quaternary ammonium salt. However, a primary amine is obtained as a major product by taking large excess of ammonia. The order of reactivity of halides with amines is RI > RBr > RCl.
APPEARS IN
RELATED QUESTIONS
Arrange the following in increasing order of their basic strength :
C6H5 – NH2, C6H5 – CH2 – NH2, C6H5 – NH – CH3
Arrange the following in increasing order of their basic strength :

How will you convert Cl−(CH2)4−Cl into hexan-1, 6-diamine?
Arrange the following in increasing order of their basic strength:
C2H5NH2, (C2H5)2NH, (C2H5)3N, C6H5NH2
Arrange the following in increasing order of their basic strength:
CH3NH2, (CH3)2NH, (CH3)3N, C6H5NH2, C6H5CH2NH2
Account for the following:
Gabriel phthalimide synthesis is preferred for synthesising primary amines.
Illustrate the following reactions giving suitable example in each case
Ammonolysis
Answer in one sentence.
Arrange the following amines in increasing order of boiling points.
n-propylamine, ethylmethyl amine, trimethylamine.
Arrange the following compounds in increasing order of their boiling points.
Ethyl alcohol, Ethyl amine, Ethanoic acid, Ethane
Among the following isomeric amines, an amine having highest boiling point is:
A compound Z with molecular formula \[\ce{C3H9N}\] reacts with \[\ce{C6H5SO2Cl}\] to give a solid, insoluble in alkali. Identify Z.
Assertion: N-Ethylbenzene sulphonamide is soluble in alkali.
Reason: Hydrogen attached to nitrogen in sulphonamide is strongly acidic.
Account for the following:
N-ethylethanamine boils at 329.3 K and butanamine boils at 350.8 K, although both are isomeric in nature.
The hydrogen bond is shortest in
Which one of the following compounds will liberate CO2, when treated with NaHCO3?
Arrange the decreasing boiling point.
\[\ce{CH3COOH, C2H5OH, CH3NH2, CH3OCH3}\]
