Advertisements
Advertisements
Question
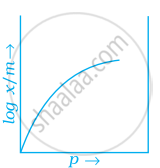
Which of the following curves is in accordance with Freundlich adsorption isotherm?
Options
Advertisements
Solution

Explanation:
Freundlich, in 1909, gave an empirical relationship between the quantity of gas adsorbed by unit mass of solid adsorbent and pressure at a particular temperature.
`x/m= kp^(1/n) (n > 1)`
The adsorption varies directly. Where `x/m` = mass of gas adsorbed per unit mass of adsorbent particle p = Pressure of gas at particular temperature.
APPEARS IN
RELATED QUESTIONS
What is an adsorption isotherm?
Describe Freundlich adsorption isotherm.
In reference to Freundlich adsorption isotherm, write the expression for adsorption of gases on solids in the form of an equation.
Answer in one sentence:
Write an equation for Freundlich adsorption isotherm.
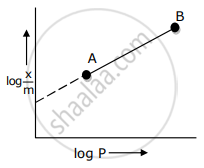
Adsorption of a gas follows Freundlich adsorption isotherm. x is the mass of the gas adsorbed on mass m of the adsorbent. The plot of log `"x"/"m"`· versus log P is shown in the given m graph. `"x"/"m"` is proportional to
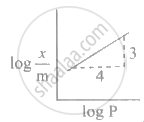
Freundlich's equation for adsorption of gas on solid is represented as ____________.
In Freundlich's adsorption isotherm, when log `("x"/"m")` is plotted against log P, slope of the graph is ____________.
Freundlich adsorption isotherm is given by the expression `x/m = kp^(1/n)` which of the following conclusions can be drawn from this expression.
(i) When `1/n` = 0, the adsorption is independent of pressure.
(ii) When `1/n` = 0, the adsorption is directly proportional to pressure.
(iii) When n = 0, `x/m` vs p graph is a line parallel to x-axis.
(iv) When n = 0, plot of `x/m` vs p is a curve.
Adsorption of gases on the solid surface is generally exothermic because.
Langmuir adsorption isotherm works particularly well.
In the freundilch adsorption isotherm, the value of x/m is 0.4 under a pressure of 0.2 at m calculate the value of the interact
Which of the following statement is INCORRECT regarding the adsorption of a gas on the surface of the solid?
In Freundlich adsorption isotherm, slope of AB line is:
The mass of gas adsorbed, x per unit mass of adsorbed, m was measured at various x pressures p. A graph between `log "x"/"m"` and log p gives a straight line with slope equal to 2 and the intercept equal to 0.4771. The value of `"x"/"m"` at a pressure of 4 atm is ______.
[Given log 3 = 0.4771]
Adsorption of a gas follows Freundlich adsorption isotherm. x is the mass of the gas adsorbed on mass m of the adsorbent. The x plot of log `x/m` versus log p is shown in the given graph. `x/m` is proportional to ______.
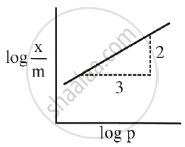
For Freundlich adsorption isotherm, a plot of log (x/m) (Y-axis) and log p (x-axis) gives a straight line. The intercept and slope for the line is 0.4771 and 2, respectively. The mass of gas, adsorbed per gram of adsorbent if the initial pressure is 0.04 atm, is ______ × 10-4 g. (log 3 = 0.4771)