Advertisements
Advertisements
Question
When a mixture of gases X and Y is compressed to 300 atm pressure and then passed over a catalyst consisting of a mixture of zinc oxide and chromium oxide (heated to a temperature of 300°C), then an organic compounds Z having the molecular formula CH4O is formed. X is a highly poisonous gas which is formed in appreciable amounts when a fuel burns in a limited supply of air; Y is a gas which can be made by the action of a dilute acid on an active metal; and Z is a liquid organic compound which can react with sodium metal to produce hydrogen gas.
(a) What are X, Y and Z?
(b) Write a balanced chemical equation of the reaction which takes place when X and Y combine to form Z. Indicate the conditions under which the reaction occurs.
Advertisements
Solution
(a) X is carbon monoxide gas (CO); it is formed when a fuel burns in limited supply of air. Y is hydrogen gas (H2); it is formed by the action of dilute acid on active metal. Z is methanol (CH3OH); it is a liquid organic compound that can react with sodium metal to produce hydrogen gas.
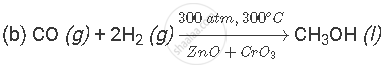
APPEARS IN
RELATED QUESTIONS
What does the symbol (aq) represent in a chemical equation?
Balance the following chemical equation:
MnO2 + HCI → MnCI2 + CI2 + H2O
Give one example of a chemical reaction characterised by formation of a precipitate.
State one characteristic of the chemical reaction which takes place when lemon juice is added gradually to potassium permanganate solution.
Why do we need to balance chemical equations?
Write word equation for the following skeletal equation:
\[\ce{AgBr -> Ag + Br2}\]
Balance the following equation. Also name the product formed.
`"H"_2 +"CI"_2 → "HCI"`
Balance the following equation. Also name the product formed.
CO2 + C → CO
Write the chemical equation for the following word equation and balance them.
Calcium + Nitrogen → Calcium nitride
Balance the following equation:
Zn + HNO3 `→` Zn(NO3)2 + H2O + NO2
Write the balanced chemical equation of the following reaction.
aluminium sulphate + sodium hydroxide → sodium sulphate + sodium meta aluminate + water.
\[\ce{MnO2 + 4HCl -> MnCl2 + 2H2O + Cl2}\]
0.02 moles of pure MnO2 is heated strongly with conc. HCl. Calculate mass of MnO2 used.
1 g of copper powder was taken in a China dish and heated. What change takes place on heating? When hydrogen gas is passed over this heated substance, a visible change is seen in it. Give the chemical equations of reactions, the name and the color of the products formed in each case.
Balance the following simple equation:
Al + O2 → Al2O3
Balance the following simple equation:
NO + O2 → NO2
Complete the given chemical reaction.
\[\ce{CuSO_{4(aq)} + Fe_{(s)} ->}\]______ + ______
What are reactants? Explain with the help of example.
Balance the given chemical reaction as per the instructions below.
\[\ce{NaOH + H2SO4 -> Na2SO4 + H2O}\]
Make a list of the elements in the chemical equation.
Reema took 5ml of Lead Nitrate solution in a beaker and added approximately 4ml of Potassium Iodide solution to it. What would she observe?

Which of the following is the correct observation of the reaction shown in the above set up?
